IVDR - taken on together.
You might be aware that your current supplier is discontinuing its niche products and now you are looking for alternatives?
Tecan's current portfolio has been reviewed to ensure compliance and a seamless transition of available products for clinical laboratories. All IBL International CE-IVDD products have been classified according to the classification rules laid out in the IVDR allowing sales during the accepted transition period. Contact us to receive the latest version of our catalog for an overview of the portfolio!
Availability of Immunoassays after May 26, 2022
We are continually working with our Notified Body on the certification of products.
Autoimmunity
Catecholamines / Neurotransmitters
Endocrinology
Saliva Diagnostics
- Cortisol Saliva Luminescence Immunoassay (30221150), 96 tests
- Cortisol Saliva Luminescence Immunoassay Bulk (30221151), 960 tests
- Cortisol Saliva ELISA (RE52611)
- 17beta-Estradiol Saliva Luminescence Immunoassay (RE62141), 96 tests
- 17beta-Estradiol Saliva Luminescence Immunoassay (RE62149), 960 tests
- 17beta-Estradiol Saliva ELISA (30121045)
- Estriol High Sensitive Saliva ELISA (30121046)
- Progesterone Saliva ELISA (RE52281)
- Melatonin direct Saliva ELISA (RE54041)
- Testosterone Saliva ELISA (RE52631)
- Testosterone Saliva Luminescence immunoassay (30191568/30191569)
The current Tecan portfolio has been reviewed in order to ensure compliance and seamless product offering to the clinical laboratories. All IBL International CE-IVDD products have been classified according to IVDR, to allow for sales during the accepted transition period. Please contact us to receive the latest version of our catalog.
New IVDR transition period
The IVDR has been in effect since May 2017 and was originally scheduled to come into force on May 26, 2022. However, the European Commission published Regulation EU 2022/112, amending the IVDR to extend the transition period for the Introduction of the IVDR.
One main point is that products that have been CE-IVDD marked prior to May 26, 2022, can continue to be manufactured and sold for an extended period but only if no significant changes are made to the intended use or design. Despite this extended transition period, certain IVDR requirements will apply to all in vitro diagnostic devices, including those that continue to be CE IVDD labeled, beginning May 26, 2022. These requirements include market surveillance and post-market surveillance of the product, vigilance, registration of economic operators and registration of devices.
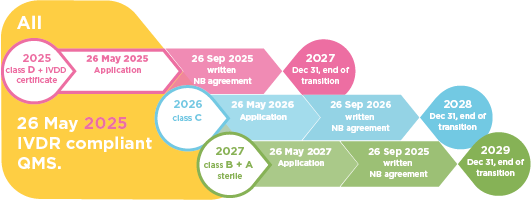
Why should clinical labs care? Here are the 5 main things you need to know:
- Know when you need to act: Even though it is not immediately necessary to shift from “IVDD” to “IVDR” devices, laboratories should keep an eye on the end of transition periods for the different device classes.
- Start early: Laboratories need to consider that devices that are not yet certified under the IVDR will need to undergo Notified Body assessment in the near future. Notified Body assessments are lengthy and complex processes.
- Stay informed: For those devices that are not certified under IVDR yet, the laboratory can request the transition plans from the manufacturers.
- Be on track with IVDD product changes: IVDD Products can be produced and sold after 26 May 2022, however no significant change in design or intended purpose is possible. If the product is changed it can only be sold with an IVDR certificate. The manufacturers now have to take a longer “time-to-market” into consideration for modified or new products.
- Don’t panic: In general, when the laboratory switches from IVDD devices to IVDR, there are no additional user requirements. If the device is the same (only changed certification), there is no need for a new validation.
Are you ready for a new chapter?
The In Vitro Diagnostics Regulation (IVDR) is the new regulatory framework to ensure the safety and performance of in vitro diagnostic devices on the European market. The IVDR replaced former EU Directive on In vitro Diagnostic Medical Devices (IVDD 98/79/EC) and came into effect May 26, 2022.
This also means that the majority of all in vitro diagnostic medical devices previously placed on the market in compliance with the IVDD have to be recertified according to the new requirements. Manufacturers must demonstrate compliance with the new IVDR requirements.
The IVDR introduced a risk-based classification system, which requires the involvement of a Notified Body in the approval of class A sterile through D, while class A non-sterile IVDs remain self-declared and D representing products with the highest risk to patients and public health. It is expected that approximately 90% of all IVDs on the market will be evaluated by a Notified Body. In comparison, this has previously applied to less than 15% of IVDs under the IVDD
Another feature is a new system for unambiguous product identification of IVDs, known as the Unique Device Identification Number (UDI), which will be used in the documentation, registration and on the finished product.
The UDI is intended to facilitate the traceability of products within the supply chain and to enable a fast and efficient recall of IVDs that pose a safety risk, as well as provide visibility and transparency for the customer.
What does it mean for laboratories/customers?
First things first: You as a customer or user are not responsible for implementing the legal framework and requirements for the IVD. Any product you find on the market with a CE marking after May 2022 will comply with the legal requirements. However, this does not mean that all products whose IVDR-compliant technical documentation has not yet been finalized will disappear after May 26, 2022, but that there will be a transition period to sell products after that date and to bridge supply needs. The complex regulatory process, conditions and rules are summarized in Article 110 of the IVDR. Some IVDs with Directive certificates issued by Notified Bodies under the Directive can still be sold and placed on the market until Dec 31, 2027 when all additional conditions are met (QMS, formal application lodged, NB agreement signed). Not all manufacturers have an agreement with a Notified Body, which is a requirement for CE-IVDR devices (Class B, C, D and A sterile), and not all Notified Bodies are designated for the IVDR (lack of market access).
Regulation beats innovation!
The market will change and with it the product range. This could possibly also affect products that you as a customer buy for your daily routine in your laboratory. As a supplier of routine and niche parameters, Tecan is interested in continuing to develop innovative products particularly for rare diseases. Tecan is at the forefront of IVDR product certification. Together with our Notified Body we have already successfully obtained IVDR certification for numerous products, and many more are in the pipeline.





