Neopterin ELISA
- Regulatory Status
- EU: CE, CDN: IVD
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 1.5 h, 1 x 10 min
- Standard range
- 1.35 - 111 nmol/L
- Specimen / Volumes
- 20 µL serum, plasma, urine
- Substrate / isotope
- TMB 450 nm
The Neopterin ELISA is intended for the quantitative determination of Neopterin in human serum, plasma (EDTA) and urine in adults.
The Neopterin ELISA is intended for the quantitative determination of Neopterin in human serum, plasma (EDTA) and urine in adults. Elevated neopterin levels can be measured as physiological marker for the indication of inflammation in the body, immune system activation and response activity. Neopterin is an unspecific marker for immune activation or other disturbances of the immune system and can be applied as an aid for early diagnosis of immune reaction like viral infection or allograph rejection. The Neopterin ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. Test results may be calculated from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges). The test kit is intended for manual use and can be adapted to different ELISA processors like Thunderbolt, DSX and BEP2000. The test kit is intended for professional laboratory use by trained personnel. The test kit is not for self-testing. The Neopterin ELISA is NOT intended for near patient testing.
is a small molecule with a low molecular weight (253.21 g/mol) and belongs to the group known as pteridines. It is synthesized upon a cellular immune reaction. When macrophages and dendritic cells are stimulated by interferon-ɣ, neopterin is released. Neopterin values reflect the entire immunological processes for monocytes/ macrophages and dendritic cells and can be seen as a global immune response marker.
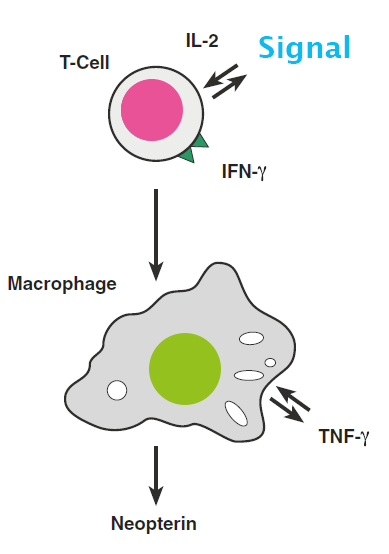
Figure 1: Release of Neopterin during cellular immune reaction [1].
An increase in neopterin concentration marks the onset of an infection in the body before antibodies are present. As soon as the immune system is triggered by interferon-ɣ, neopterin is released by macrophages.
Neopterin is related to:
Insights and cues from neopterin about the pathology of SARS-Cov-2: Synthesized upon stimulation of macrophages and dendritic cells, neopterin is an early, well-characterized biochemical marker of an ongoing immune reaction to viral infection [2]. There is evidence that neopterin levels correlate with the severity of COVID-19 progression and may be prognostic in COVID-19 patients [3–6]. The Neopterin ELISA kit sensitively detects this small molecule prior to clinical symptoms of infection and thus, may be a tool for early detection of SARS-CoV-2 infection.
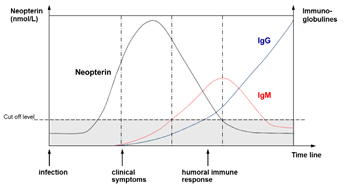
Figure 2: Schematic time course of neopterin concentration in blood in relation to the rise and fall of antibody presence in response to viral infection [2].
Measurement in bodily fluids: The determination of neopterin levels in human body fluids offers a useful and innovative tool to monitor diseases associated with the activation of cell-mediated immunity. The ELISA is validated for measuring neopterin in serum, plasma (EDTA) and urine. Neopterin has higher stability in body fluids which makes sample handling and measurement easier compared to other cytokines. The low molecular weight enables neopterin molecules to pass rapidly through the intravascular area, where it is released into urine after glomerular filtration. The half-life in the human body is only affected by renal excretion. As a non-invasive method, urinary Neopterin to creatinine ratio determination is also helpful in monitoring disease progression and the effects of therapies, as well.
Increasing neopterin levels in various infections precede the clinical manifestation and seroconversion. Normally samples are not tested for all possible infections. Therefore, the measurement of neopterin in blood donor samples is a useful tool in order to reduce the risk of infections via blood transfusion [2].
Potential other diagnostic applications:
Neopterin indicates a plethora of infections before the onset of symptoms and research is investigating neopterin levels and Covid-19 [6].
Innate immune activation during Covid-19 infection is associated with pernicious clinical outcomes.

Figure 3: Neopterin is a biomarker of fatal outcome in SARS-CoV-2 infected patients [6].
The authors investigated the course of patients from the time symptoms occurred to discharge or death. The survival curves for Covid-19 patients (above) indicate that elevated neopterin levels (>53nm) correlated with a lower probability of survival, while lower levels (<53nM) in serum concentration led to a higher chance of survival.

Figure 4: Distinction between healthy and Covid-19 patients. Red symbols represent deceased patients. Blue symbols represent patients who recovered within one month-follow-up (survivors) [6].
A principal component analysis (PCA) was applied and neopterin levels in serum were measured with the Tecan Neopterin ELISA. In order to determine the effect of some combinations of comorbidities the PCA was generated, which would allow for stratification of the patients according to fatal clinical outcome. This principal component analysis allowed to make a clear distinction between Covid-19 patients who recovered from those who died.
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. CE IVD under IVDR is to be launched soon. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.