Acetylcholine Receptor-Ab (ARAb) RRA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 100
- Method
- RRA
- Incubation time
- 1 x 2 h, 1 x 30 min
- Standard range
- 0 - 8 nmol/L, cut-off 0.25 nmol/L
- Specimen / Volumes
- 20 µL serum, plasma
- Substrate / isotope
- 125I < 100 kBq
Myasthenia gravis is an autoimmune disease which can cause severe muscle weakness and greatly impact quality of life. Diagnosis can be difficult, but state-of-the-art disease biomarkers and targeted assays are available to increase the likelihood that a patient with myasthenia gravis is diagnosed early and can be treated appropriately. In myasthenia gravis, the body’s own immune system produces antibodies that attack the skeletal muscles responsible for breathing and various other movements. Patients typically experience muscle weakness following activity that improves with rest. Early and accurate diagnosis of myasthenia gravis is important because, despite the absence of a cure, there are effective treatments that can help control symptoms and improve a patient’s quality of life.
The Acetylcholine autoantibody radio receptor assay is intended for the semi-quantitative determination of autoantibodies against the acetylcholine receptor in adult human serum and EDTA plasma. The measurement of highly specific autoantibodies against the acetylcholine receptor (AChr) is appropriate as an aid for diagnosis of myasthenia gravis (MG), a rare but long-term muscle disease leading to muscle weakness of varying severity. In diagnostics, the determination of autoantibodies against the acetylcholine receptor is used as the first screening tool in patients with suspected Myasthenia Gravis disease, as in around 80% of MG patients autoantibodies against AChR are detectable (seropositive MG). Furthermore determination of autoantibodies provides information concerning (patho-) physiological state of the patient. For definition of myasthenic subgroup, such as early-onset MG, late-onset MG, thymoma-associated or MuSK-associated MG, further diagnostic investigation is necessary. Subgroups will not be defined using this assay.
Acetylcholine receptor from human muscle is used as antigen in this radio receptor assay. The receptors are labelled with 125I-alpha-bungarotoxin, which binds the receptors most specifically and almost irreversible. Autoantibodies present in the patient´s serum or plasma attach to the labelled receptors. The resulting immune complexes are precipitated with anti-human IgG. The amount of radioactivity of the sediment is directly proportional to the concentration of acetylcholine receptor autoantibodies of the sample. The assay requires general purpose laboratory instruments and consumables such as gamma counter, vortexer and pipettes to execute the test. The manual kit assay test instructions must be strictly adhered to and verified by the laboratory. Test results are calculated from a standard curve and compared to defined cut-off. The test kit is intended for professional laboratory use by trained personnel. The test kit is not for home or lay person use.
The measurement of highly specific autoantibodies against the acetylcholine receptor (AChr) supports the diagnosis of myasthenia gravis (MG), a rare but long-term muscle disease leading to muscle weakness of varying severity. The muscles of the eyes, face and for swallowing are most commonly affected. In diagnostics, the determination of autoantibodies against the acetylcholine receptor is used as the first serological tool in patients with suspected myasthenia gravis disease.[1-3]
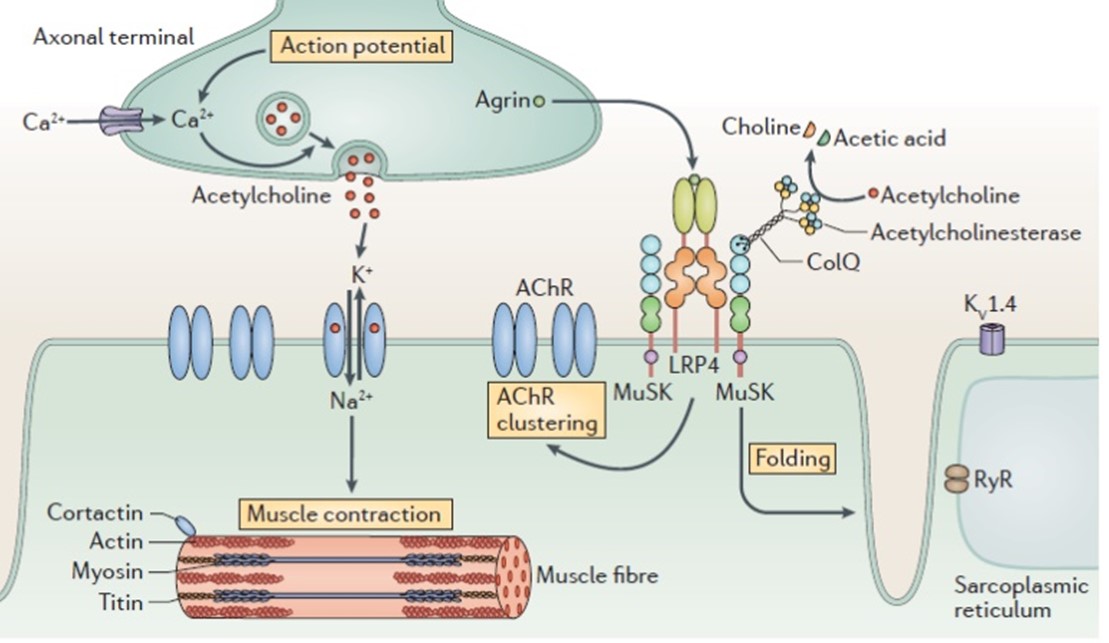
Fig.1: Neuromuscular junction in myasthenia gravis (MG). [11]
AChR antibodies are directly pathogenic through crosslinking of AChRs following accelerated receptor degradation. Furthermore pathogenic function by inducing AChR conformational changes or blocking acetylcholine binding can be observed.[1] A distinction is made between several forms of myasthenia gravis, for example generalized myasthenia gravis or ocular myasthenia gravis. Autoantibodies against AChR are detectable in serum of at least 80 % of patients with generalized myasthenia gravis[2,4] and in 50-60 % of patients with ocular myasthenia.[5] D-penicillamine can cause anti-AChR and anti-MuSK antibody-positive MG, a rare phenomenon, which is reversed after discontinuation of D-penicillamine treatment.[6,7] The examination of patients with suspected MG disease is usually performed with multiple antibody determinations. In the case of AChR seronegativity, the presence of other antibodies such as those against MuSK or Lrp4 can support diagnosis of an existing MG disease in AChR-seronegative patients.[8]
The most sensitive and specific AChR antibody test is a radioimmunoassay (binding antibody assay) using human AChR. It has been the gold standard for MG diagnosis for many years due to its high specificity and sensitivity.[9,10]
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
[1] Gilhus, N. E., & Verschuuren, J. J. (2015). Myasthenia gravis: subgroup classification and therapeutic strategies. The Lancet. Neurology, 14(10), 1023–1036. https://doi.org/10.1016/S1474-4422(15)00145-3
[2] Binks, S., Vincent, A., & Palace, J. (2016). Myasthenia gravis: a clinical-immunological update. Journal of neurology, 263(4), 826–834. https://doi.org/10.1007/s00415-015-7963-5
[3] Farmakidis, C., Pasnoor, M., Dimachkie, M. M., & Barohn, R. J. (2018). Treatment of Myasthenia Gravis. Neurologic clinics, 36(2), 311–337. https://doi.org/10.1016/j.ncl.2018.01.011
[4] Vincent, A., & Newsom-Davis, J. (1985). Acetylcholine receptor antibody as a diagnostic test for myasthenia gravis: results in 153 validated cases and 2967 diagnostic assays. Journal of neurology, neurosurgery, and psychiatry, 48(12), 1246–1252. https://doi.org/10.1136/jnnp.48.12.1246
[5] Nicolle M. W. (2016). Myasthenia Gravis and Lambert-Eaton Myasthenic Syndrome. Continuum (Minneapolis, Minn.), 22(6, Muscle and Neuromuscular Junction Disorders), 1978–2005. https://doi.org/10.1212/CON.0000000000000415
[6] Poulas, K., Koutsouraki, E., Kordas, G., Kokla, A., & Tzartos, S. J. (2012). Anti-MuSK-and antiAChR-positive myasthenia gravis induced by d-penicillamine. Journal of Neuroimmunology, 250(1-2), 94-98.
[7] Li, Mingqiang, et al. "Clinical characteristics of AChRAb and MuSKAb double seropositive myasthenia gravis patients." Clinical neurology and neurosurgery 172 (2018): 69-73.
[8] Huda, S., Woodhall, M. R., Vincent, A., & Heckmann, J. M. (2016). Characteristics Of acetylcholine‐receptor‐antibody–negative myasthenia gravis in a South African cohort. Muscle & nerve, 54(6), 1023-1029.
[9] Lazaridis, K., & Tzartos, S. J. (2020). Autoantibody specificities in myasthenia gravis; implications for improved diagnostics and therapeutics. Frontiers in Immunology, 11, 212.
[10] Wang, S., Breskovska, I., Gandhy, S., Punga, A. R., Guptill, J. T., & Kaminski, H. J. (2018).Advances in autoimmune myasthenia gravis management. Expert review of neurotherapeutics, 18(7), 573–588. https://doi.org/10.1080/14737175.2018.1491310
[11] Gilhus, N. E., Skeie, G. O., Romi, F., Lazaridis, K., Zisimopoulou, P., & Tzartos, S. (2016). Myasthenia gravis—autoantibody characteristics and their implications for therapy. Nature reviews neurology, 12(5), 259-268.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.