The first IVDR sIL-2R ELISA providing trustworthy results for aiding in the sarcoidosis diagnosis
Request your trial kit now!
The sInterleukin-2-Receptor ELISA is intended for the quantitative determination of human soluble IL-2 receptor (sIL-2R) in serum and plasma (EDTA, citrate, heparin).
sIL-2R levels can be used as physiological marker for the indication of inflammation in the body, in-vivo immune system activation and response activity. As such the measurement of sIL-2R levels can be applied as an aid in the diagnosis or monitoring of therapy of autoimmune diseases (e.g. sarcoidosis).
The sInterleukin-2-Receptor ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the on the sandwich principle measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. Test results may be calculated from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges). The test kit is intended for manual use and can be adapted to different ELISA processors like EVOlyzer.
The test kit is intended for professional laboratory use by trained personnel. The test kit is not for self-testing. The sInterleukin-2-Receptor ELISA is NOT intended for near patient testing.
Features of the ELISA
- calibrated against NIBSC 97/600 standard preparation
- sufficient reagents for adaptation to different ELISA processors
- low and high kit control
- results reported in arbitrary U/mL
- first IVDR ELISA (based on Eudamed database search on Feb 23, 2024)
Your benefits
- Trustworthy results: >95% specificity compared to established IVD solutions (N=216 from 7 different labs)
- Scalable solution: manual assay that can be easily automated (e.g. Thunderbolt®, DSX®, EVOlyzer®)
Furthermore, the kit correlates very well with the previous sIL-2R ELISA (30166211) as shown below in the linear regression graph.
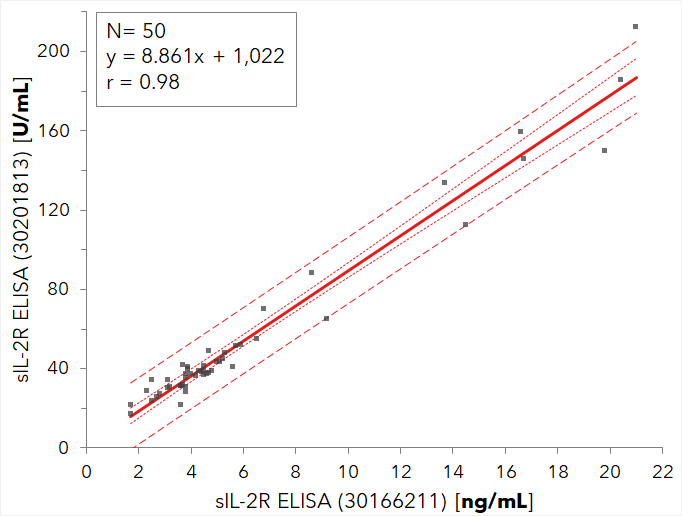
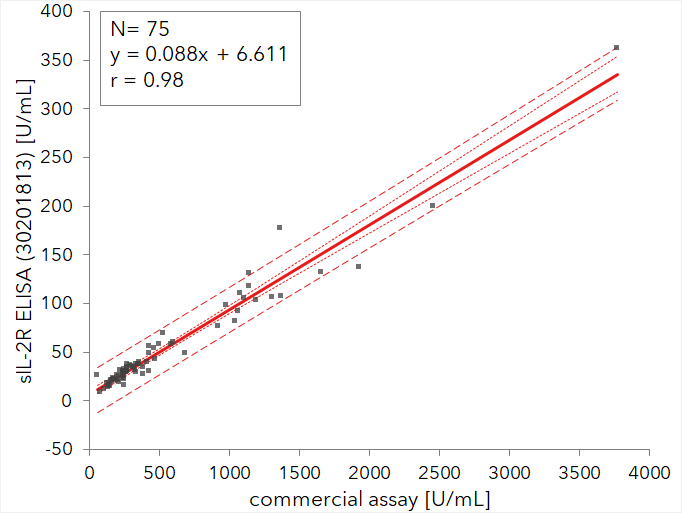
In a blinded external comparison study the correlation to a similar IVD device was examined. The results indicate an excellent comparability as shown with the linear regression graph below and with the correlation coefficient of r=0.98.
Summary on the interleukin-2 receptor and its function
The interleukin-2 receptor (IL-2R) is a monomeric, heterodimeric or -trimeric protein¹ expressed on the surface of certain immune cells like lymphocytes that binds and responds to a cytokine called IL-2². Interleukin-2 (IL-2) is a body own signaling molecule mainly produced by activated T-cells. It acts on different types of B- and T-cells via the Interleukin-2 Receptor. The functional Interleukin-2 Receptor consists either of two or three protein chains. The dimeric IL-2R consists of the IL-2Rβ (CD122) chain and the IL-2Rγ (CD132) that form an intermediate affinity receptor for IL-2. If the IL-2α (CD25) chain is incorporated into the receptor complex the IL-2R attains high affinity for IL-2³.
In 1985 the soluble Interleukin-2 Receptor (sIL-2R, sCD25, sTAC, sIL2R, IL-2RA) was first described as being actively released by activated peripheral blood T-cells via proteolytic cleavage of the cell surface IL-2R⁴.
Clinical relevance of analyte with applicable population
Measuring levels of sIL-2R in adults are considered as a good parameter to determine on-going immune response, which can be used to monitor immune mediated diseases⁵.
This property is taken into account by several clinical guidelines. The German Society of Nephrology (Deutsche Gesellschaft für Nephrologie) together with the German Clinical Chemistry and Laboratory Society (Deutsche Gesellschaft für Klinische Chemie und Laboratoriumsmedizin e. V. (DGKL)) recommend to measure sIL-2R together with other parameters in the diagnostic work-up of acute kidney failure to rule out a sarcoidosis origin⁶. Furthermore, the German Ophthalmologic Society (Deutsche Ophthalmologische Gesellschaft e.V.) and the Professional Association of Ophthalmologists in Germany (Berufsverband der Augenärzte Deutschlands e.V.) recommend in their clinical guidelines for optic neuritis (Optikusneuritis) also the measurement of sIL-2R to rule out a sarcoidotic background⁷. Another clinical guideline for the diagnostic of polyneuropathies from the German Society of Neurology (Deutsche Gesellschaft für Neurologie) also recommends to measure sIL-2R, if there is an indication of pulmonary involvement⁸. The guidelines refer to two additional publications⁹,¹⁰ explaining how to use sIL-2R measurements. In particular, conditions that are characterized by excessive production of lymphocytes, termed lymphoproliferative disorders show high sIL-2R levels compared to healthy controls. But also granulomatous diseases, in which T-cell activation is a typical hallmark, show elevated sIL-2R levels.
If you want to learn more about the use of sIL-2R in a laboratory setting, please click on the picture below to watch a webinar hosted by Dr. Bob Meek, St. Antonius Ziekenhuis, NL.
















