Early and accurate measurement of Vitamin B12 deficiency
is essential for preventing the occurrence of irreversible neurological dysfunction. IBL International offers the Active-B12 (holotranscobalamin) ELISA for measuring Vitamin B12 which is bound to transcobalamin (TC), the only biologically active fraction that can be delivered to cells. Less than 30% of the total Vitamin B12 in plasma circulates as Active-B12 (HoloTC). The remaining 70% are bound to haptocorrin (HoloHC). Because changes in HoloTC levels occur earlier than Total-B12 in Vitamin B12 depletion, it constitutes a faster way to prove Vitamin B12 deficiency. Independent clinical studies show the improved clinical diagnostic value obtained with measuring Active-B12 compared to assessing the Total Plasma Vitamin B12 concentration. The Active-B12 (holotranscobalamin) ELISA does not require pre-analytical steps, which is the main source of laboratory variation.
Improved diagnosis of Vitamin B12 deficiency
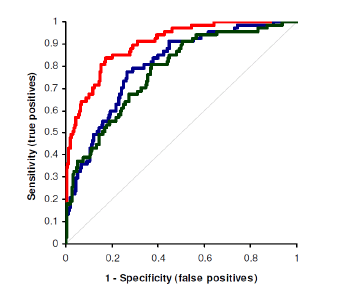
An independent study showed that Active-B12 (HoloTC) is a more sensitive and more specific biomarker for assessing Vitamin B12 deficiency than Total-B12 and Methylmalonic Acid (MMA).
IBL International Active-B12 (HoloTC) ELISA outcome algorithm
Subjects at risk of B12 deficiency
*Please note that leading experts in the Vitamin B12 field recommend a cut-off value of 35pmol/L for most populations (Herrmann W., Obeid R., Schorr H., Geisel J. The usefulness of holotranscobalamin in predicting Vitamin B12 status in different clinical settings. J Curr Drug Metab. 2005; 6(1):47-53.)
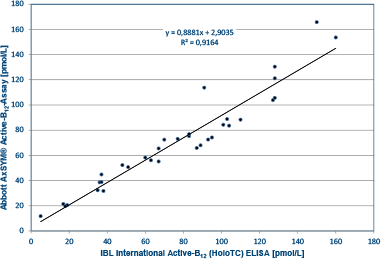
External comparison study shows excellent correlation between IBL International’s Active-B12 (HoloTC) ELISA and the Abbott AxSYM® Active-B12-Assay
Excerpt from the Instructions for Use
Enzyme immunoassay for the quantitative determination of holotranscobalamin (HoloTC) in human serum.
Vitamin B12 deficiency is a major public health issue; it is widespread, but the elderly are the group most at risk. It is estimated that 10-15% of over-65’s will have B12 deficiency. Neurological symptoms may be the only sign of deficiency, and can be irreversible if treatment is delayed. The only biologically-active fraction that can be delivered to cells is Vitamin B12 attached to transcobalamin (TC). Less than 30% of the Vitamin B12 in plasma circulates as Active-B12 (HoloTC). The remaining 70% or so is bound to haptocorrin. Accurate measurement of Vitamin B12 status is needed because of the high prevalence and serious complications of deficiency. Total plasma Vitamin B12 concentration is the current standard clinical test for Vitamin B12 deficiency, but it has various limitations: in particular, most of the Vitamin B12 measured is the haptocorrin-bound fraction. This suggests a prima facie clinical benefit from measuring Active-B12, in terms of remedying the poor sensitivity and specificity of Total-B12 measurement.
For concrete data please consult the Instruction for Use in the download box on the top right side.














