Chromogranin A New Gen. RUO ELISA
- Regulatory Status
- RUO
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1x 1 h; 1x 2 h; 1x 10 min
- Standard range
- 60 - 1,000 ng/mL
- Specimen / Volumes
- 20 µL Serum, Plasma
- Substrate / isotope
- TMB 450 nm
| instructions for use |
|---|
| instructions for use |
|---|
is the most important general marker for neuroendocrine tumors. This marker is often elevated in people with functionally active as well as inactive tumors. Unlike other biological markers, chromogranin A is relatively stable and largely correlates with the tumor mass. Tecan has been offering a Chromogranin A ELISA kit for many years.
All other parameters have been optimized to best meet the requirements. Incubation times have been reduced, dilution and recovery tests have been improved, the robustness of the kit has been optimized.
The new kit uses the same antibodies as the previous test to maintain the same specificity. The antibodies are now produced in cell culture, thus reducing the use of animals.
The experiment was performed on two batches of Chromogranin A New Gen. ELISA and 2 batches of Chromogranin A RIA-CT on 109 serum samples covering the whole calibration range.
The results below show a good correlation between the kits with a slope close to 1.0 which will facilitate the switch from radioimmunoassay to ELISA.
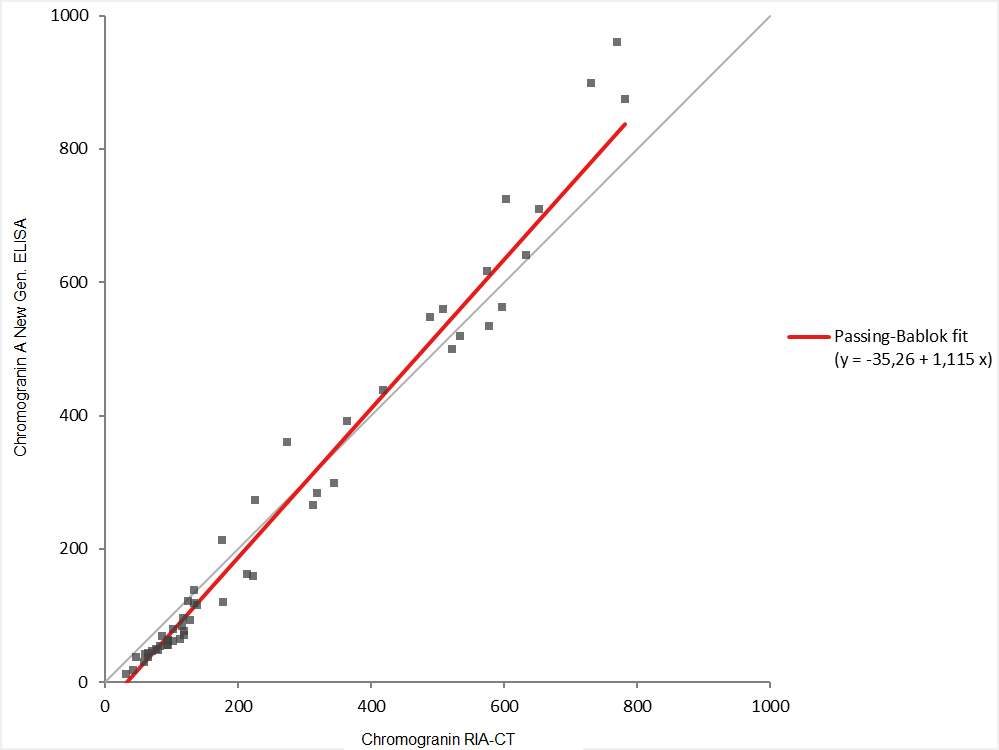
Similar correlation was performed using 53 samples covering the whole range of concentrations. The little gap on the slope is linked to the adjustment of the new kit to obtain optimal dilution performances along the calibration curve. Nevertheless, the correlation is good.

Hook effect: No hook effect observed up to 2 million ng/mL.
Recovery and Dilution: To control the similarity of the assay response between exogenous CGA and serum CGA, recovery tests were performed. The results show a perfect similarity of the assays whatever the origin of the antigen hence meeting Cisbio internal standards in force (90%-110%).
Out-of-range samples must be diluted. To ensure that dilutions do not compromise the accuracy of the results provided, cascade dilution tests were performed (1/2; 1/4, 1/8, 1/16). The results show very good dilution performances with a recovery close to 1.0, a slope interpolation close to 1.0 and a correlation coefficient also close to 1.0.
Biotin interferences: No significant interference up to biotin concentrations of 600 ng/mL.
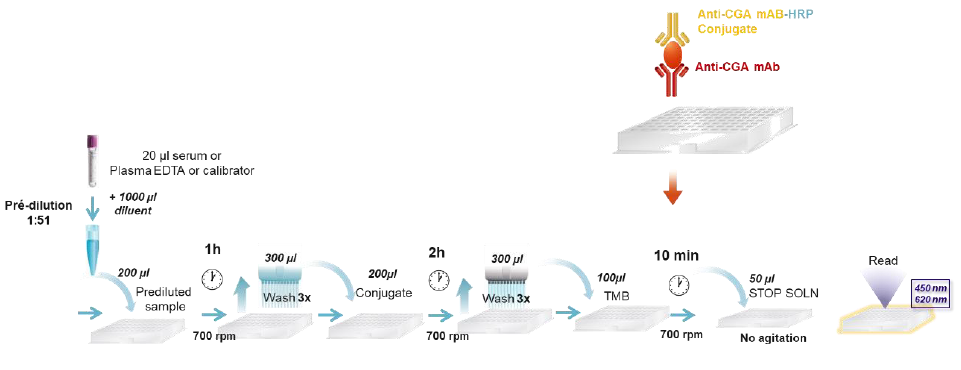
The assay procedures has also been simplified and a short scheme is provided below. Please refer to the instructions for use for details.
Product manufactured by CisBio, and distributed by Tecan, IBL International GmbH.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.