Periostin ELISA
- Regulatory Status
- RUO
- Kit size
- 12x8
- Method
- ELISA
- Incubation time
- 1 x O/N, 1 x 90 min., 1 x 10 min.
- Standard range
- 0 - 2000 ng/mL
- Specimen / Volumes
- 5 µL serum, plasma, cell culture supernatant, nasal lavage fluid, vitreous fluid, tears, BALF
- Substrate / isotope
- TMB 450nm






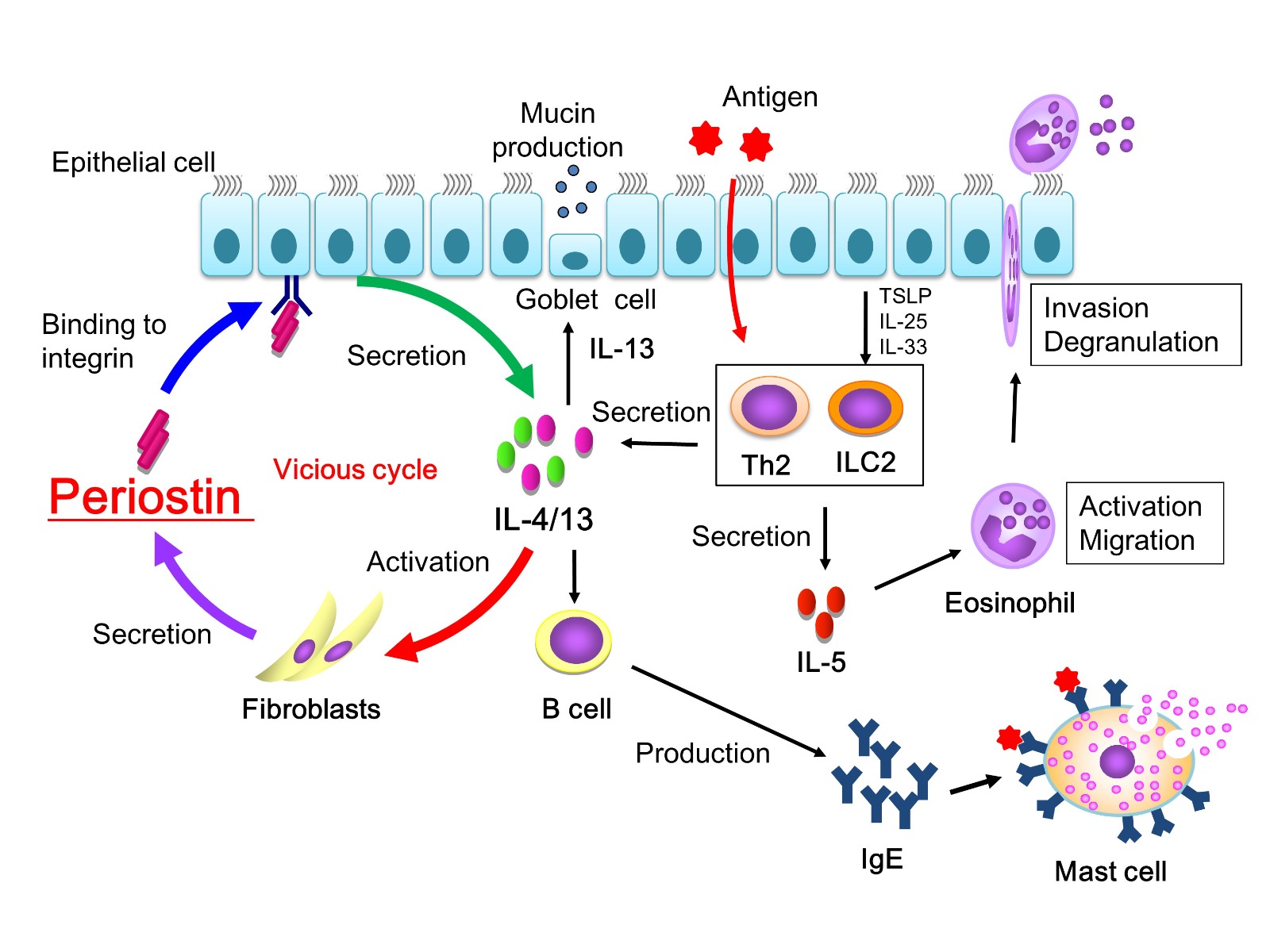 Fig. 1 Periostin as an inducer of a vicious cycle in the IL-4/Il-13 release
Fig. 1 Periostin as an inducer of a vicious cycle in the IL-4/Il-13 release





