17ß-Estradiol Saliva ELISA
- Regulatory Status
- IVD
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 2 x 1 h, 1 x 30 min
- Standard range
- 2 - 64 pg/mL
- Specimen / Volumes
- 50 µL Saliva
- Substrate / isotope
- TMB 450 nm
Enzyme immunoassay for the quantitative measurement of 17beta-Estradiol in human saliva.
17β-Estradiol (Estradiol; E2) is the most important natural estrogen. It is present in females and males. In the former, the estrogens stimulate the growth of sex organs and the development of secondary sexual characteristics, and they also affect the gonadotropin secretion. 17β-Estradiol measurements are useful to detect estrogen deficiency in delayed puberty, primary and secondary amenorrhea and in postmenopausal women mostly.
The new IBL International 17ß-Estradiol Saliva ELISA is calibrated to the LC-MS/MS reference method and includes harmonized reagents within the IBL Saliva ELISA product line.
Benefits of the 17ß-Estradiol Saliva ELISA
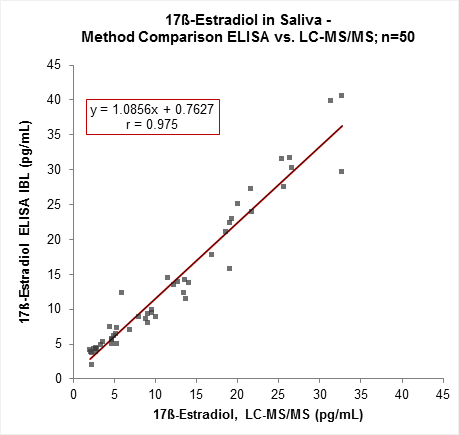
Our Saliva ELISA product line (Testosterone, Progesterone, 17ß-Estradiol, Estriol, DHEA) can be processed in a similar way and be easily performed in combination:
Estradiol is secreted into circulation by the ovaries, placenta, adrenal gland, and testes or is produced by extra-glandular conversion of secreted androgen precursors. Estradiol is biologically the most active of the naturally produced human estrogens. In the postmenopausal women, Estradiol originates from extra-glandular conversion of androgens and circulates in low, non-cyclic concentrations.The changes in the hormone levels are registered during the different the phases of the menstrual cycle.
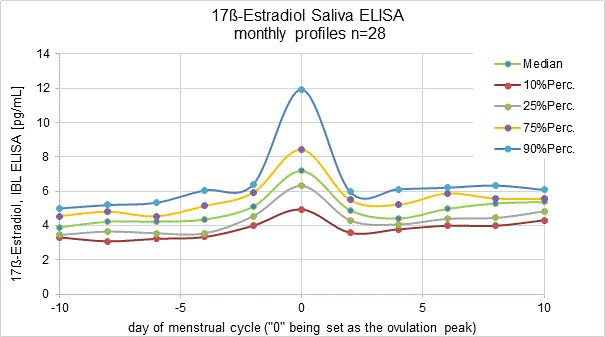
Similar to Testosterone and Progesterone, 17ß-Estradiol also shows short time pulsating dynamics, especially in females. Therefore single saliva determinations will result in arbitrary values. We recommend collecting 3 to 5 saliva samples within 2 hours. In the laboratory, equal volumes of the individual saliva samples can be mixed. This mixed sample results in a mean 17ß-Estradiol value, which represents the active hormone concentration in a reproducible way.
In women, the estrogens stimulate the growth of sex organs and the development of secondary sexual characteristics, and they also affect the gonadotropin secretion. In the males, the role of 17β-Estradiol is less well defined although it seems to be involved in the regulation of gonadotropin secretion. In non-pregnant women 17β-Estradiol is almost exclusively produced by ovary. Especially after having changed into the menopause, estrogens are produced by the liver, the brain, the muscles and by the adipose tissue, too. Moreover, the measurement of 17β-Estradiol is helpful to determine a lack of estrogens, which may be expressed as delayed puberty, primary and secondary amenorrhoea, and occurs in the menopause. Other hormone assays are required for proper interpretation and differential diagnosis. The concentration of gonadotropin should be measured to find the origin of the lack of estrogens (synthesis or regulation). The determination of 17β-Estradiol is useful for the diagnosis of pubertas praecox of girls. Concerning men, the level of 17β-Estradiol may be used for the differential diagnosis of gynecomastia.
Being principally bound to sex hormone binding globulin (SHBG) and to serum albumin, only 1 - 3 % of estradiol circulating in plasma is present in its free form. Only this portion represents the active part in the endocrine regulation. The free hormone is released in equal amounts in saliva.
Therefore measuring the 17ß-Estradiol in saliva is a convenient non-invasive method. Easier sample collection without repeated venipunctures and reduced stress on the specimen offer benefits to the patient while sensibility and accuracy of the results are ensured.
For concrete data please consult the Instruction for Use in the download box on the top right side.
[2] Greenspan, F. S., & Gardner, D. G. (2001). Basic and clinical endocrinology.
[3] Gavrilova, N., & Lindau, S. T. (2009). Salivary sex hormone measurement in a national, population-based study of older adults. Journals of Gerontology Series B: Psychological Sciences and Social Sciences, 64(suppl_1), i94-i105.
[4] Mendoza, K., Curran, M., Salimetrics, L. L. C., & Lindau, S. T. (2007). Salivary Estradiol Measurement in Wave I of the Social Life Health & Aging Project. J Gerontol B Psychol Sci Soc Sci. 2009 Nov; 64B(Suppl 1): i94–i105. doi: 10.1093/geronb/gbn028
[5] Tivis, L. J., Richardson, M. D., Peddi, E., & Arjmandi, B. (2005). Saliva versus serum estradiol: implications for research studies using postmenopausal women. Progress in Neuro-Psychopharmacology and Biological Psychiatry, 29(5), 727-732.
[6] Jameson JL, De Groot LJ (25 February 2015). Endocrinology: Adult and Pediatric E-Book. Elsevier Health Sciences. p. 2179. ISBN 978-0-323-32195-2.
[7] Shirtcliff, E. A., Dahl, R. E., & Pollak, S. D. (2009). Pubertal development: correspondence between hormonal and physical development. Child development, 80(2), 327-337.
[8] Fernández-Garcia, B., Lucia, A., Hoyos, J., Chicharro, J. L., Rodriguez-Alonso, M., Bandres, F., & Terrados, N. (2002). The response of sexual and stress hormones of male pro-cyclists during continuous intense competition. International journal of sports medicine, 23(08), 555-560.
[9] Ikegami, S., Moriwake, T., Tanaka, H., Inoue, M., Kubo, T., Suzuki, S., ... & Seino, Y. (2001). An ultrasensitive assay revealed age‐related changes in serum oestradiol at low concentrations in both sexes from infancy to puberty. Clinical endocrinology, 55(6), 789-795.
[10] Kang, J. Y., Park, J. Y., Chun, S. I., Suh, H. S., Lee, K., & Ahn, R. S. (2014). Puberty-related changes in cortisol, dehydroepiandrosterone, and estradiol-17β secretions within the first hour after waking in premenarcheal girls. Neuroendocrinology, 99(3-4), 168-177.
[11] McIver, B., Romanski, S. A., & Nippoldt, T. B. (1997, December). Evaluation and management of amenorrhea. In Mayo Clinic Proceedings (Vol. 72, No. 12, pp. 1161-1169). Elsevier.
[12] Buckler, H. (2005). The menopause transition: endocrine changes and clinical symptoms. British Menopause Society Journal, 11(2), 61-65.
[13] Goletiani, N. V., Keith, D. R., & Gorsky, S. J. (2007). Progesterone: Review of safety for clinical studies. Experimental and clinical psychopharmacology, 15(5), 427.
[14] Bowen R (2000-08-06). "Placental Hormones". Retrieved 2020-29-12. http://www.vivo.colostate.edu/hbooks/pathphys/reprod/placenta/endocrine.html
[15] Hall, J. E. (2004). Neuroendocrine control of the menstrual cycle. In J. F. Strauss, III, & R. L. Barbieri (Eds.), Yen and Jaffe’s reproductive endocrinology: Physiology, pathophysiology, and clinical management (5th ed., pp. 195–211). Philadelphia: Elsevier
[16] Cable, J. K., & Grider, M. H. (2020). Physiology, Progesterone. StatPearls
[17] Frank, G. R. (2003). Role of estrogen and androgen in pubertal skeletal physiology. Medical and pediatric oncology, 41(3), 217-221.
[18] Herrera et al., 2017: Herrera, A. Y., Hodis, H. N., Mack, W. J., & Mather, M. (2017). Estradiol therapy after menopause mitigates effects of stress on cortisol and working memory. The Journal of Clinical Endocrinology & Metabolism, 102(12), 4457-4466
[19] Vermeulen, A., Kaufman, J. M., Goemaere, S., & Van Pottelberg, I. (2002). Estradiol in elderly men. The aging male, 5(2), 98-102.
[20] Dielen, C., Fiers, T., Somers, S., Deschepper, E., & Gerris, J. (2017). Correlation between saliva and serum concentrations of estradiol in women undergoing ovarian hyperstimulation with gonadotropins for IVF/ICSI. Facts, views & vision in ObGyn, 9(2), 85.
[21] Lu, Y. C., Bentley, G. R., Gann, P. H., Hodges, K. R., & Chatterton, R. T. (1999). Salivary estradiol and progesterone levels in conception and nonconception cycles in women: evaluation of a new assay for salivary estradiol. Fertility and sterility, 71(5), 863-868.
[22] Bacon, J. L. (2017). The menopausal transition. Obstetrics and Gynecology Clinics, 44(2), 285-296.
[23] Gandara, B. K., Leresche, L., & Mancl, L. (2007). Patterns of salivary estradiol and progesterone across the menstrual cycle. Annals of the New York Academy of Sciences, 1098, 446.
[24] Schmidt, J., Wenzel, F., & Blessing, F. (2020) Practical evaluation of two commercial immunoassays for the quantification of steroid hormones in human saliva. Journal of Cellular Biotechnology, (Preprint), 1-9.
[25] Shirtcliff, E. A., D. A. Granger, et al. (2000). "Assessing estradiol in biobehavioral studies using saliva and blood spots: simple radioimmunoassay protocols, reliability, and comparative validity." Horm Behav 38(2): 137-47
[26] Quaiser‐Pohl, C., Jansen, P., Lehmann, J., & Kudielka, B. M. (2016). Is there a relationship between the performance in a chronometric mental‐rotations test and salivary testosterone and estradiol levels in children aged 9–14 years?. Developmental psychobiology, 58(1), 120-128.
[27] Choe, J. K., Khan-Dawood, F. S., & Yusoff-Dawood, M. (1983). Progesterone and estradiol in the saliva and plasma during the menstrual cycle. American journal of obstetrics and gynecology, 147(5), 557-562
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.