Dehydroepiandrosterone (DHEA) Saliva ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 2 h, 1 x 30 min
- Standard range
- 20 - 2160 pg/ml
- Specimen / Volumes
- 50 µL saliva
- Substrate / isotope
- TMB 450 nm
is intended for the quantitative measurement of the DHEA hormone secreted by the adrenal gland using human saliva sample from adult patients. This test is intended for the measurements of DHEA levels as a physiological marker as an aid in the diagnosis and treatment of suspected dysfunction of the adrenal gland, in conjunction with other clinical parameters, such as Cortisol and other hormones. The information in addition to other clinical observations and diagnostic test is useful in assessing the level of adrenal function as a determination of physiological status in aging adults. Measurement of DHEA levels together with other hormones has been shown to be useful in the evaluation of hormone imbalance that results in various clinical conditions like hyperandrogenism (excess of androgen hormones), e.g. polycystic ovary syndrome (PCOS). Use of non-invasive sample types from saliva make the test suitable for frequent sampling and testing of DHEA levels.
The DHEA Saliva ELISA is NOT appropriate for use in the diagnosis and treatment of DHEA-secreting adrenal carcinomas. The DHEA Saliva ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. The assay is adaptable by laboratory personnel to automate on open ELISA based liquid handler platforms; however, the programming of the steps and timing required by the manual kit assay test instructions must be strictly adhered to and verified by the laboratory. Test results may be calculated manually from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges). The test kit is intended for professional laboratory use by trained personnel. The test kit is not for home or lay person use.
The IBL International DHEA Saliva ELISA correlates very well to the LC-MS/MS method, and is providing specified reference values for women, men, age and time of the day.
Benefits of the measurement of DHEA in saliva and for the IBL DHEA Saliva ELISA
*Valid lot numbers of TMB, stop solution and wash solution are stated on the kits outer labels and Quality Control certificates.
The DHEA concentration of 140 saliva samples from apparently healthy females and males (morning and evening samples) were measured in the DHEA Saliva ELISA. The DHEA level decreases in elderly females and males as well as in evening samples. Due to the pulsatile dynamics of DHEA secretion, repeated saliva samples were analysed:
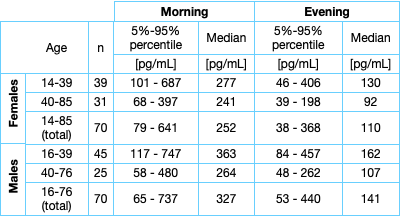
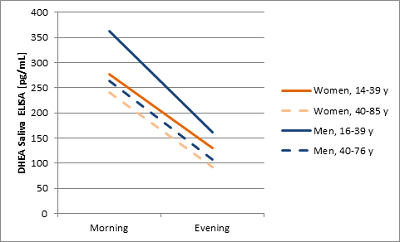
It is recommended that each laboratory establishes its own range of normal values. As several specific biases, like age, gender and time of the day need to be taken into account, it is recommended that each laboratory should establish their own reference range for their patient population.
To assess the adrenal function of a patient, it is necessary to measure a baseline DHEA value dependent on the individual or affected population and the time of the day, and compare to a healthy control group population. Multiple measurements are recommended to establish a patients profile. Measurement of DHEA levels together with other hormones has been shown to be useful in the evaluation of hormone imbalance that results in various clinical conditions like hyperandrogenism (excess of androgen hormones), e.g. polycystic ovary syndrome (PCOS). In the specific case of PCOS, DHEA levels in salivary samples were identified to be For hormone imbalance that results in various clinical conditions like polycystic ovary syndrome (PCOS, a type of Hyperandrogenism) values of 390-690 pg/mL, with values up to 1190 pg/mL were identified. As measurements of DHEA levels as a physiological marker as an aid in the diagnosis and treatment of suspected dysfunction of the adrenal gland, has to be done in conjunction with other clinical parameters, such as Cortisol and other hormones, the results themselves should not be the only reason for any therapeutic consequences. DHEA should not be considered as sole parameter to consider for establishing a diagnostic, and as such it is important to incorporate information from other adrenal hormones values for a complete clinical diagnostic picture. The results alone should not be the only reason for any therapeutical consequences. They have to be correlated to other clinical observations and diagnostic tests.
Summary and explanation
DHEA (Dehydroepiandrosterone) and its sulphate ester DHEA-S, as C-19 steroid hormones, belong to the group of androgens [1]; [2]. The zona reticularis of the adrenal glands produces approximately 80% of DHEA, with the remainder produced by the testes, ovaries, and brain [3]. DHEA serves as a precursor in testosterone and estrogen synthesis [1]; [2]. In blood DHEA is loosely bound to albumin and SHBG [4]. Only a small portion of hormone, approximately 5%, stays unbound and can penetrate saliva via intracellular diffusion and ultrafiltration mechanisms and, therefore, salivary concentrations reflect serum concentrations [5]. As such, it can be concluded that DHEA level in saliva samples reflects the circulating level of free bioactive steroid fraction in the body, rather than total circulating levels that are measured in blood, which are confounded by the presence of circulating binding proteins. DHEA levels show significant diurnal variation which is dependent on adrenocorticotrophic hormone (ACTH) [2]. DHEA has been shown to display a flat pattern of secretion after waking, followed by a progressive decline to 3 hours post-awakening with no significant change thereafter [6]. The normal DHEA concentration changes significantly with age. The level of DHEA starts to increase at the adrenarche and reaches its maximum at the age of 20-30 years. After that the adrenal production of DHEA decline in both males and females, until concentrations of DHEA return to pre-adrenarche levels in persons over 80 years old [7]. The DHEA Saliva ELISA assay is intended for adult patients only. Biochemical/physiological adult pattern can be achieved already with 14 years of age, at least for girls and 16 years for boys [8]. There is an evidence of decreasing level of DHEA and increased Cortisol/DHEA ratio to be involved in the cognitive and immune impairments in elderly people, as well as influencing the ability of older people to perform daily tasks. Higher Cortisol/DHEA ratios, rather than the hormone levels independently, have been associated with immune impairments and infection risk in older adults exposed to chronic physical and psychological stress [6]. Acute Stress and Posttraumatic Stress Disorder are characterized by elevated level of DHEA in comparison to control groups, due to increased ACTH secretion initiated by stress and/or traumatic events [9-13]. Testing DHEA in saliva suggests non-invasive and stress-free sample collection method, which allows to avoid additional stress burden on the patients with psychiatric disorders [13]. Use of non-invasive sample types from saliva make the test ideal for frequent sampling and testing of DHEA levels. The key laboratory findings that help a clinician to evaluate the HPA axis status in an individual are total cortisol output, cortisol awaking response, cortisol diurnal rhythm, total DHEA(S) and Cortisol/ DHEA ratio [9]. As such, the measurement of DHEA levels in saliva, in addition to other clinical observations and diagnostic test is useful in assessing the level of adrenal function as a determination of physiological status in aging adults. Measurements of DHEA levels are useful as an aid in the diagnosis and treatment of adrenal disorders in conjunction with measurements of other sex steroids like Testosterone, DHT and Androstenedione. Hirsutism, acne, alopecia, oligo-amenorrhea and polycystic ovary syndrome (PCOS) are clinical expressions of hyperandrogenism [14; 15; 16]. The usage of saliva provides a sensitive, simple, reliable and non-invasive diagnostic approach evaluating for biochemical hyperandrogenemia [14].
Pulsatile dynamics
Due to the pulsatile dynamics of DHEA secretion repeated saliva sampling is recommended. It is recommended to collect 5 samples within 2 hours after awakening time. In the laboratory, equal volumes of the individual saliva samples can be mixed. This mixed sample results in a mean free DHEA value, which represents the active hormone concentration in a reproducible way.
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. CE IVD under IVDR to be launched soon. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.