Dehydroepiandrosterone (DHEA) ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 1 h; 1 x 30 min
- Standard range
- 0.37 - 30 ng/mL
- Specimen / Volumes
- 20 µL Serum
- Substrate / isotope
- TMB 450 nm
Intended Use
Enzyme immunoassay for the quantitative determination of Dehydroepiandrosterone (DHEA) in human serum.
Intended Purpose
Enzyme immunoassay for the quantitative determination of Dehydroepiandrosterone (DHEA) in human serum.
This test is intended for the measurement of DHEA levels in human serum as a physiological marker of suspected dysfunction of the adrenal gland in conjunction with additional observations. The measurement of DHEA levels serve as an aid of diagnosis for hormone imbalances that results in various clinical conditions like premature adrenarche, hyperandrogenism (excess of androgen hormones) like polycystic ovary syndrome (PCOS) or Congenital adrenal hyperplasia (CAH) and people affected by virilization or hirsutism.
The test kit is intended for manual use and can be adapted to different ELISA processors like EVOlyzer. The test kit is intended for professional use. The test kit is not for home or layperson use.
Clinical significance
Dehydroepiandrosterone (DHEA) is a C19 steroid also known as 5-androsten-3â-ol-17-one.[1] The adrenal cortex is the primary source of circulating concentrations of DHEA and DHEA-S of the adrenals.[2]
DHEA is synthesized in two steps from the precursor molecule cholesterol, which is converted to Pregnenolone and finally to DHEA, while DHEA-S is either converted through DHEA or cholesterol.[3]
The synthesis occurs differently in women and men, while in healthy women, the synthesis of DHEA and DHEA-S occurs exclusively in the adrenal cortex; in men, it is estimated that 5 % of DHEA-S and 10 % to 25 % of the circulating DHEA are secreted by the testes. Both DHEA and its sulfated metabolite DHEA-S are endogenous hormones, which are secreted by the adrenal cortex in response to adrenocorticotrophin (ACTH). The adrenal cortex is the primary source of circulating concentrations of DHEA and DHEA-S, with DHEA-S being the most abundant product of the adrenals. Once in circulation, DHEA-S can be metabolized back to DHEA by sulfohydrolases in peripheral and adrenal tissues. It was reported that 64 % and 74 % of the daily production of DHEA-S is converted to DHEA in women and men but only about 13 % of DHEA is metabolized back to DHEA-S. DHEA and DHEA-S serve as the precursors to approximately 50 % of androgens in adult men, 75 % of active estrogens in pre-menopausal women, and almost 100 % of active estrogens after menopause.[1]
DHEA level display a flat pattern after waking, followed by a progressive decline three hours post-awakening with no significant change thereafter.[5] Basal DHEA levels can be observed in the morning (8 a.m.) and males show higher DHEA level than females or younger adult but higher levels than older people. In older people the DHEA levels decline and the circadian rhythm is flattened.[5]
Morning DHEA or DHEA-S levels are higher than bedtime levels, however, it is highly dependent on when the first morning sample is collected. The closer to time of waking, the higher the levels of the morning sample will be when compared to bedtime samples (50 - 100 % higher).[6] The key
laboratory findings that help a clinician to evaluate the HPA axis status is the individual cortisol level, which follows a rhythm, total DHEA and Cortisol/ DHEA ratio.[7]
The normal DHEA concentration changes significantly with age. The level of DHEA starts to increase at the adrenarche and reaches its maximum at the age of 20 - 30 years. Later in life the adrenal production of DHEA declines in both men and women, until concentrations of DHEA return to pre-adrenarche levels in people over 80 years.[8]
Measurements of DHEA levels is applied as an aid in the diagnosis of adrenal dysfunctions e.g. congenital adrenal hyperplasia (CAH) in conjunction with measurements of other sex steroids like testosterone, DHT and androstenedione. Hirsutism, acne, alopecia, oligo-amenorrhea and polycystic ovary syndrome (PCOS) are clinical expressions of hyperandrogenism.[9-11]
A disorder resulting from a deficiency of one of five enzymes required for cortisol synthesis in the adrenal cortex. 21-Hydroxylase deficiency is the most common cause for congenital adrenal hyperplasia (CAH). The severe deficiency results in the salt-losing form and in case of a partial deficiency it results in virilizing. A lifelong glucocorticoid supplementation to reduce excess levels of adrenal androgens are necessary to manage this disorder consists of DHEA levels show significant diurnal variation, which is dependent on adrenocorticotrophic hormone (ACTH).[12; 13]
LC-MS as our benchmark
Dehydroepiandrosterone (DHEA) and its sulfate ester DHEA-S are the most abundant sex steroid hormones in humans, providing a large precursor reservoir for the synthesis of androgens and estrogens. However, only the non-sulfated (free) DHEA shows biological activity.
Tecan IBL offers a highly specific ELISA for determining serum DHEA levels, which was calibrated against the reference method LC-MS /MS. The assay's distinguishing features are:
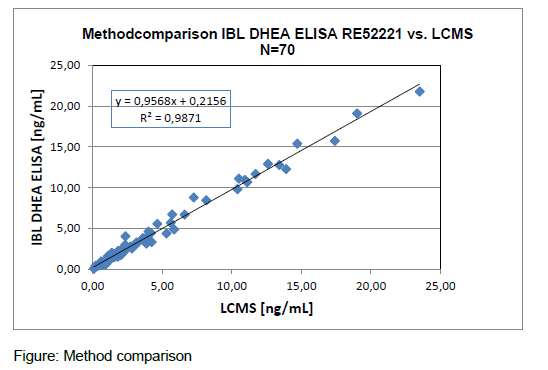
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.