1,25-dihydroxy Vitamin D ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 18h, 1h, 15min
- Standard range
- 3 - 180 pg/mL
- Specimen / Volumes
- 500 µL Serum
- Substrate / isotope
- TMB 450 nm
The quantification of the physiologically active Vitamin D metabolite 1,25-dihydroxy Vitamin D is a necessary diagnostic tool for several pathological conditions such as:
The quality of the measuring method, which our assay fulfills a hundred percent, therefore is indispensable.
The levels of 1,25-dihydroxy Vitamin D in plasma or serum is 100 to 1000-fold less than that of 25-hydroxy Vitamin D. Due to its low concentrations and the presence of many similar metabolites, the exact measurement of 1,25-dihydroxy vitamin D still requires to do an extraction and separation step with organic solvents of the sample prior any measurement system. This extraction with organic solvents still today is the gold standard extraction method.
"The successive abandonment of sample extraction, chromatography, […] in immunoassays has undoubtedly contributed to the inconsistencies reported by Farrell et al, […]." (Graham Carter, DEQAS organizer, Carter GD. Clin Chem. 2012;58(3):486-8.)
The extraction suppresses the potential interference from sample matrix, heterophilic antibodies and abnormal concentrations of Vitamin D binding proteins. And the additional separation on solid phase cartridges further suppresses the interference from other Vitamin D metabolites such as 25-hydroxy Vitamin D, 24,25-dihydroxy Vitamin D, 3-epi-25-hydroxy Vitamin D and 25,26-dihydroxy Vitamin D.
This leads to good correlation of our ELISA with the gold standard technology LC-MS/MS.
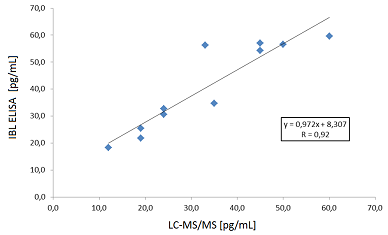
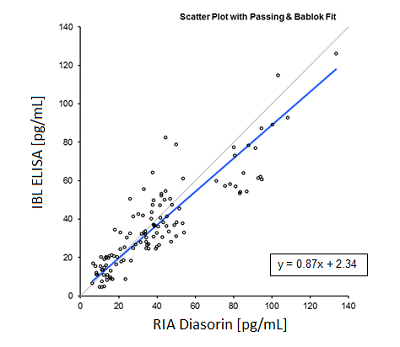
Furthermore the ELISA was also correlated to the still widely used radioimmunoassay from Diasorin. Also here the ELISA showed a good fit.
General information
1,25(OH)2 Vitamin D is the active form of Vitamin D with regard to the known functions whereas 25OH Vitamin D and Vitamin D itself can be excluded as being physiologically functional. 1,25(OH)2 Vitamin D stimulates the intestinal absorption of both calcium and phosphorus. It also stimulates bone resorption and mineralization thereby preventing the development of rickets and osteomalacia. 1,25(OH)2 Vitamin D is also active in other tissues responsible for Calcium transport (placenta, kidney, mammary gland,...) and endocrine glands such as parathyroid glands. 1,25(OH)2 Vitamin D is rapidly metabolized and its half life in plasma is approximately 12h. Its is further metabolized to either calcitroic acid, essentially without any biological activity, or via 24-hydroxylation to 1,24,25-trihydroxy vitamin D. This compound has less biological activity than its parent and this metabolic route is considered as a minor pathway. The levels of 1,25(OH)2 Vitamin D in plasma or serum is 100- to 1000-fold less than that of 25OH Vitamin D. Due to its low concentrations and the presence of many similar metabolites, the measurement of 1,25(OH)2 Vitamin D requires extraction and separation by chromatography.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.