IGF-I ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 1h (RTS), 1 x 30 min (RTS), 1 x 15 min
- Standard range
- 2-50 ng/mL
- Specimen / Volumes
- 10 µL Serum or Plasma
- Substrate / isotope
- TMB, 450 nm
The measurement of insulin-like growth factor-I (IGF-I) in serum is of especially high utility in the diagnostic work-up of growth disturbances, particularly with regard to human growth hormone (hGH) deficiency or acromegaly. The most important advantage of measuring IGF-I over hGH are its stable circadian levels, i.e. even a single measurement is of conclusive significance.
In view of the above, measurement of IGF-I should be put on the front burner in laboratory diagnostic work-up.
The advantages of our ELISAs are:
IBL International´s IGF-I enzyme linked immunosorbent assay (ELISA) by IBL International is used for measuring IGF-I in human serum/plasma, cerebrospinal fluid and other body fluids as well as in conditioned cell culture media of many human cell lines.
What is more, the assay can also be used for the measurement of IGF-I in other mammals such as primates, cattle, pig, sheep, horse, donkey, goat, dog, cat, rabbit and guinea pig. However, the kit is not suited for rat, mouse and chicken samples
Insulin-like growth factors I and II (IGF-I, IGF-II) play a key role in proliferation, differentiation and specific functions of many cell types. IGF-I is identical to somatomedin C (Sm-C) and is regulated by the growth hormone (hGH/GH) and nutrition. Unlike many other peptide hormones, IGFs are bound to specific binding proteins (IGFBP) with a high affinity. A substantial problem in the measurement of IGF-I levels is the interference by IGFBPs. When measured in untreated serum samples, inaccurate results are obtained due to the extremely slow dissociation of the IGF-I/IGFBP-3 complex during assay incubation. For this reason, only a fraction of IGF-I is available for measurement. Interferences are determined by the IGF-I to IGFBP ratio in the sample.

In order to circumvent this problem, a simple assay was developed in which samples are acidified and diluted using a buffer of a special composition prior to performing ELISA.
Apart from hGH, a number of other factors affect serum IGF-I levels. Low values are found in malnutrition/malabsorption, hypothyroidism, liver disease, untreated diabetes mellitus, chronic inflammatory diseases, malignancies and multiple trauma. Elevated levels, on the other hand, are found in precocious puberty and obesity.
In order to be able to correctly interpret IGF measurements, it is of utmost importance to consider the age-dependent pattern of IGF-1 levels, which you can find in the Instruction for Use.
| Percentiles | |||||
| Pubertäts stadium | 0.1th | 5th | 50th | 95th | Pubertal Stage |
| 1 | 61 | 105 | 186 | 330 | 1 |
| 2 | 85 | 156 | 298 | 568 | 2 |
| 3 | 113 | 196 | 352 | 631 | 3 |
| 4 | 171 | 268 | 431 | 693 | 4 |
| 5 | 165 | 263 | 431 | 706 | 5 |

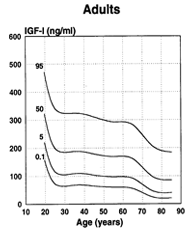
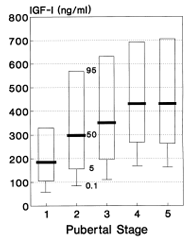
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.