Heparin Binding Protein ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 2 x 1h, 1 x 10min
- Standard range
- 12.5 - 200 ng/mL
- Specimen / Volumes
- 100µL plasma
- Substrate / isotope
- TMB 450nm
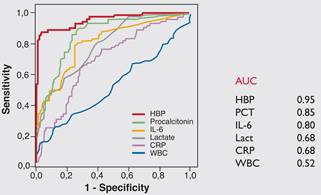
Enzyme immunoassay for the quantitative determination of Heparin Binding Protein in human plasma. Heparin Binding Protein is a new marker that has demonstrated utility in identifying patients at risk of developing severe sepsis. Heparin Binding Protein, also known as CAP37 and azurocidin, is synthesised in neutrophils. Once released from activated neutrophils, it induces a rearrangement of the endothelial cell cytoskeleton, resulting in increased permeability of the endothelium. At the site of infection, HBP is responsible for the recruitment and activation of monocytes and other inflammatory mediators. It is also internalised by monocytes to prolong survival and enhance cytokine production. HBP therefore directly contributes to the maintenance and progression of inflammation. A publication in 2009 demonstrated that in febrile patients presenting to the Emergency Department (ED), there is a close correlation between increased plasma HBP levels and the development of severe sepsis with hypotension or shock. In this prospective study of 233 febrile adult patients with suspected sepsis, Heparin Binding Protein was shown to be the best predictor of severe sepsis with Receiver-operating characteristic (ROC) plot showing an Area Under Curve (AUC) of 0.95, exceeding that of Procalcitonin, IL-6 and Lactate:
 Distributed by IBL International
Distributed by IBL International
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.