SCCA2 ELISA
- Regulatory Status
- RUO
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x O/N, 1 x 90 min, 1 x 30 min
- Standard range
- 0.0063 - 0.2 ng/mL
- Specimen / Volumes
- 10 µL serum
- Substrate / isotope
- TMB 450nm
This kit utilizes an enzyme-linked immunosorbent assay (ELISA) method to quantify SCCA2 in serum. Samples are added to microtiter plate with 96-wells (solid phase) coated with anti-SCCA antibody, and after washing, a POD-conjugated anti-SCCA2 antibody is added to react, an immune complex is formed. After the unreacted POD-conjugated anti-SCCA2 antibody is removed by washing, a chromogenic substrate is added to perform the enzymatic reaction, and color develops in proportion to the amount of SCCA2 in the samples. The color development is stopped, and the color intensity is measured at 450nm.
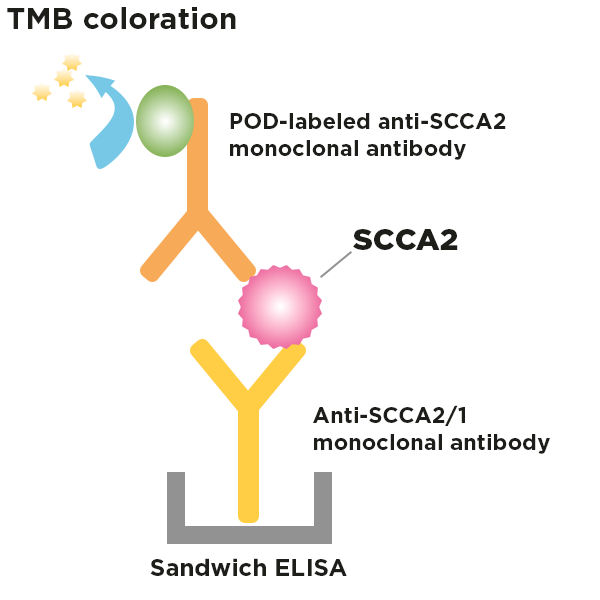
Fig.: Principal of the SCCA2 ELISA
SCCA (squamous cell carcinoma antigen) is a serine protease inhibitor belonging to serpin superfamily, which is a protein with a molecular weight of about 45 kDa produced mainly by epithelial cells. SCCA contains the isoforms SCCA1(SERPIN B3) and SCCA2(SERPIN B4, which are independent gene products transcribed from distinct loci, albeit with very high-amino-acid homologies of 91%.
Unlock deeper insights and improve sample management in dermatology research with the SCCA2 ELISA kit
It is known that interleukin 13 (IL-13), one of the type Th2 cytokines, plays crucial roles in allergic inflammation. SCCA2 was identified as one of the molecules whose expression is elevated when airway epithelial cells are stimulated with IL-4 and IL-13, cytokines that are central in allergic responses.¹ Serum SCCA2 levels in children with atopic dermatitis have been reported to be significantly elevated according to the severity of the disease, even when compared with existing blood tests²,³,⁴.
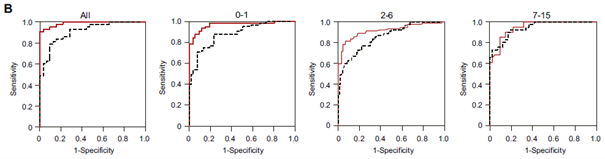
Fig. ROC curve analyses of SCCA2 (red solid line) and TARC (black dashed line) in all and <1 year old, 2 to 6 years old, and 7 to 15 years old of control subjects and AD children
In the treatment of atopic dermatitis, it is important to select and use anti-inflammatory drugs appropriately according to their severity. Currently there are more than 30 different drug candidates in clinical phase 2 and 3 trials⁵ needing reliable objective tools such as the SCCA2 ELISA for stratification of study participants and to see possible effects.
Learn more about how SCCA2 could move the understanding of atopic dermatitis from subjective to objective assessment by watching the video linked below.
Distributed by TECAN, IBL International GmbH
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.