Active-B12 (holotranscobalamin) ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 1h, 1 x 35 min., 1 x 30 min.
- Standard range
- 12 - 154 pmol/L
- Specimen / Volumes
- 100µL serum
- Substrate / isotope
- PNPP 405 nm
is essential for preventing the occurrence of irreversible neurological dysfunction. IBL International offers the Active-B12 (holotranscobalamin) ELISA for measuring Vitamin B12 which is bound to transcobalamin (TC), the only biologically active fraction that can be delivered to cells. Less than 30% of the total Vitamin B12 in plasma circulates as Active-B12 (HoloTC). The remaining 70% are bound to haptocorrin (HoloHC). Because changes in HoloTC levels occur earlier than Total-B12 in Vitamin B12 depletion, it constitutes a faster way to prove Vitamin B12 deficiency. Independent clinical studies show the improved clinical diagnostic value obtained with measuring Active-B12 compared to assessing the Total Plasma Vitamin B12 concentration. The Active-B12 (holotranscobalamin) ELISA does not require pre-analytical steps, which is the main source of laboratory variation.
The Active-B12 (Holotranscobalamin) assay is a manual enzyme-immunoassay (EIA) for the quantitative determination of holotranscobalamin (HoloTC) in human serum. HoloTC (vitamin B12 bound to transcobalamin) is used as an aid in the diagnosis and treatment of vitamin B12 deficiency, in apparently healthy individuals. For professional use only.
Three binding proteins are involved in the transport of vitamin B12 around the body - Intrinsic Factor (IF), transcobalamin (TC) and haptocorrin (HC). These binding proteins ensure the efficient uptake of the very small amounts of vitamin B12 available from the diet. When TC and HC bind vitamin B12 the resulting complexes are known as holotranscobalamin (HoloTC) and holohaptocorrin (HoloHC) to distinguish them from the proteins carrying no vitamin. The major fraction in the circulation, HoloHC, represents 70-90% of vitamin B12 in the blood but is biologically inert. HoloTC represents only 10- 30% of vitamin B12 circulating in the blood but is the only form of vitamin B12 that can be taken up by cells in the body. The TC protein alone transports vitamin B12 from its site of absorption in the ileum to tissues and cells. The vitamin is then internalised as the HoloTC (vitamin B12 bound to transcobalamin) complex via a specific receptor-mediated uptake. This process delivers vitamin B12 into the cells of the body and provides the vitamin as a co-enzyme for essential cellular functions such as DNA synthesis. As HoloTC has a shorter circulating half-life compared to HoloHC the earliest change that occurs on entering negative vitamin B12 balance is very likely to be a decrease in serum HoloTC concentration [1]. The measurement of Total Serum B12 suffers from some limitations; in particular, most of the measured cobalamin is bound to biologically inert HC. Several studies have been published which conclude that HoloTC would be a better indicator of vitamin B12 status than Total Serum B12[2], [3]. As expected, HoloTC levels are low in patients with biochemical signs of vitamin B12 deficiency [4]. Low values have been reported in vegetarians [5] , vegans [6] , and in populations with a low intake of vitamin B12 [7] . Notably, low levels of HoloTC but not Total B12 in serum were reported in patients with Alzheimer’s disease compared to levels in a healthy control group [8]. HoloTC levels reflect vitamin B12 status, independent of recent absorption of the vitamin [9].
An independent study showed that Active-B showed that Active-B (HoloTC) is a more sensitive and more specific biomarker for assessing Vitamin B deficiency than Total-B and Methylmalonic Acid (MMA). Table 1: Sensitivity and specification comparison between Active-B , Total-B and Methylmalonic Acid (MMA) for assessing Vitamin B deficiency
| Biomarker | Area under the curve | Sensitivity | Specificity |
|---|---|---|---|
| Active-B12 (HoloTC) | 0.899 | 55% | 96% |
| Total-B12 | 0.801 | 46% | 88% |
| Methylmalonsäure (MMA) | 0.776 | 81% | 63% |
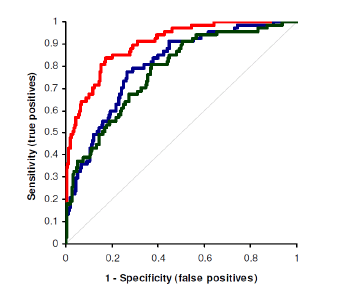
Figure: Measurement of B-12-status with the Active-B12 (HoloTC) ELISA. [2]
Enzyme immunoassay for the quantitative determination of holotranscobalamin (HoloTC) in human serum.
Vitamin B12 deficiency is a major public health issue; it is widespread, but the elderly are the group most at risk. It is estimated that 10-15% of over-65’s will have B12 deficiency. Neurological symptoms may be the only sign of deficiency, and can be irreversible if treatment is delayed. The only biologically-active fraction that can be delivered to cells is Vitamin B12 attached to transcobalamin (TC). Less than 30% of the Vitamin B12 in plasma circulates as Active-B12 (HoloTC). The remaining 70% or so is bound to haptocorrin. Accurate measurement of Vitamin B12 status is needed because of the high prevalence and serious complications of deficiency. Total plasma Vitamin B12 concentration is the current standard clinical test for Vitamin B12 deficiency, but it has various limitations: in particular, most of the Vitamin B12 measured is the haptocorrin-bound fraction. This suggests a prima facie clinical benefit from measuring Active-B12, in terms of remedying the poor sensitivity and specificity of Total-B12 measurement.
Distributed by Tecan,IBL-International.For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.