HMGB1 Express ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 2h, 1 x 1h, 1 x 20min.
- Standard range
- 2.5 - 80 ng/mL
- Specimen / Volumes
- 10 µl serum, plasma
- Substrate / isotope
- TMB 450 nm
Now available, the successor product to the well known and most cited ST51011 HMGB1 ELISA co-marketed by IBL International, part of TECAN group and Shino-Test Corp. from Japan.
The new HMGB1 Express ELISA now allows to get results quicker as before as we were able to manage to reduce working time from 2 days to 4 hours. Furthermore, the new kit is more sensitive than the ST51011 HMGB1 ELISA due to newly used monoclonal antibody resulting in more endogenous HMGB1 to be detected by the HMGB1 Express ELISA. Still the kit correlates well to the predecessor HMGB1 ELISA.

Being a Severe Acute Respiratory Syndrome disease, COVID-19 might be causing great damage to the lung and if involved with comorbidities might lead to death. Statistics openly published with media indicate that despite being on ventilation still roundabout 85% of these patients still die. A recent review shows how HMGB1 might link into the severity of the pathology of SARS-CoV-2 in the lungs.
Andersson U, Ottestad W, Tracey KJ. Extracellular HMGB1: A therapeutic target in severe pulmonary inflammation including COVID-19? Mol Med. 2020;26:42
doi: 10.1186/s10020-020-00172-4
“…The focus of this review is a possible role of the proinflammatory, endogenous mediator high mobility group box 1 protein (HMGB1) in the pathogenesis of COVID-19. …”
“…on the functional role HMGB1/TLR4-mediated neuroinflammation in brain diseases of both infectious and sterile origin. COVID-19 also emerge with features of neuroinflammation including fever, loss of small, taste and appetite. We suggest that HMGB1 contributes to these symptoms via TLR4 expression…”
“…HMGB1 transfection complexes were not toxic to cultured cells. It is thus conceivable that the RNA of SARS-CoV-2 virus might reach the cellular cytosol through HMGB1-assisted transport, as previously discussed…”
“…Chloroquine phosphate-based therapy in China and hydroxychloroquine treatment in South Korea have been reported to improve outcome in COVID-19 infections…Chloroquine has been demonstrated to decrease HMGB1 secretion from activated innate immunity cells. Chloroquine-mediated alkalinization of lysosomes inhibiting HMGB1-caused lysosomal leakage is a plausible mechanism that might be beneficial for COVID-19 treatment to prevent lysosomal leakage...”
If you want to learn more about the involvement of HMGB1 in COVID-19 pathology click on the picture below to watch a video interview with Prof. Andersson.
Over the years, HMGB1 has increasingly attracted the attention of basic as well as clinical researchers and has become one of the most intriguing molecules within the complete arsenal of proteins of the innate immune system.
IBL International’s HMGB1 ELISA, which has been developed by Shino-Test Corporation, Japan, in cooperation with Prof. Dr. Ikuro Maruyama, Kagoshima University, is the worldwide renown assay to quantify HMGB1 in any experimental setting.
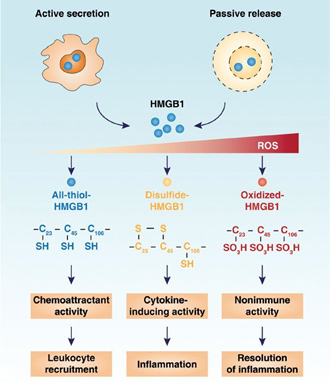 Figure: Redox control of HMGB1 activity (Tang D. et al. MolMed. 2012;18:1360-1362).
Figure: Redox control of HMGB1 activity (Tang D. et al. MolMed. 2012;18:1360-1362).
HMGB1 is a nuclear protein that can bind and bend DNA, but it can also be released to the extracellular environment where it exerts additional functions. The unraveling of the post-translational modifications has led to a better understanding of the mechanism of its translocation and its function within the immune system. It will now also allow more in-depth studies into the role of HMGB1 in numerous pathologies, including
Through its collaboration with HMGB1 experts from academia and commercial partners, IBL International is at the forefront of new developments. We at IBL offer the most complete range of products - including the highly sensitive ELISA for the quantitative measurement of HMGB1 - which have been widely used and are cited in many publications.
 Figure: HMGB1 biology by the current state of scientific knowledge (click to enlarge)
Figure: HMGB1 biology by the current state of scientific knowledge (click to enlarge)
Excerpt from the Instructions for Use
In 1973 a group of non-histone nuclear protein with high electrophoretic mobility was discovered and termed High-Mobility Group (HMG) proteins. The High Mobility Group Box1 Protein (HMGB1) ubiquitous small protein (30 kDa) that is the major component of the non-histone nuclear protein group. The protein is ubiquitously expressed in eukaryotes and hereby serves a plethora of different functions within the subcellular localizations. HMGB1 can easily shuttle between the nucleus and cytoplasm and is widely expressed in various tissues. Within the nucleus HMGB1 is involved in the DNA-binding and folding activity, as well as a regulator of various key DNA processes (recombination, repair, transcription, gene expression and genomic stability). However, once released into the cytoplasm by immunocompetent cells HMGB1 is involved in disease pathogenesis and inflammation (damage-associated molecular pattern (DAMP). It also serves as a necrosis marker and is involved in immune response processes (liver failure, rheumatoid arthritis, acute lung injury) and disseminated intravascular coagulation and measured in case of an drug-induced liver failure. Significant clinical value has HMGB1 in case of an Drug-induced liver injury (DILI). DILI, the first cause of acute liver failure, refers to liver injury caused by drugs or chemical agents e.g. Acetaminophen overdose. According to the IMI SAFE-T consortium in collaboration with DILIN and PSTC evaluated new liver safety biomarkers, which includes HMGB1 (beside osteopontin, keratin 18 and MCSFR1, miR-122 and GLDH) as i) a marker of progression prediction from hepatocellular injury to severe DILI and ii.) prediction of the occurrence of suspected intrinsic liver injury (EASL Clinical Practice Guidelines)
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.