Estriol High Sensitive Saliva ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1x2h; 1x30min
- Standard range
- 2 - 288 pg/mL
- Specimen / Volumes
- 100 µL saliva
- Substrate / isotope
- TMB 450 nm
The Estriol Sensitive Saliva ELISA will be IVDR ready soon!
Saliva Testing of hormone levels
Enzyme immunoassay for the quantitative determination of Estriol in human saliva.
The Estriol high sensitive Saliva ELISA is intended for the quantitative measurement of Estriol in human saliva.
Estriol is an estrogen typically produced during pregnancy. Estriol level found in non-pregnant women are similar to level both during pre- and post-menopause and are similar to the level in men. The estriol saliva assay is commonly applied to monitor estriol level in women who take supplements as part of a hormone replacement therapy and as an indicator of premature delivery in pregnant women. However, this assay is not intended to be used in the context of screening for congenital disorders of the fetus (such as Down Syndrome).
Additonal hormone assays are recommended for an interpretation of the level of estriol.
The Estriol high sensitive Saliva ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. The assay is adaptable by laboratory personnel to automate on open ELISA based liquid handler platforms; however, the programming of the steps and timing required must be verified by the laboratory. Test results are calculated from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges).
The test kit is intended for professional laboratory use by trained personnel. The test kit is not for self-testing. The Estriol high sensitive Saliva ELISA is not intended for near-patient testing.
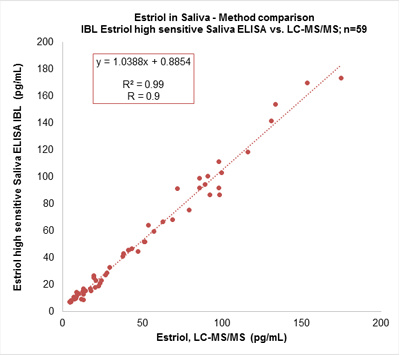
Mass spectrometry (MS) is considered as the reference for steroid hormone quantification. This methodology does however represents a large investment and as such, is not available to all laboratories. The main IBL saliva assays have been developed to correlate to this reference method, and even more importantly, only require low sample volume for quantification of the steroid hormones.
Our Saliva ELISA product line (Cortisol, Testosterone, Progesterone, 17ß-Estradiol, Estriol, DHEA) can be processed in a similar way and be easily performed in combination:
*Valid lot number of TMB, stop solution and wash solution are stated on the outer label and on the Quality Control certificates.
The IBL International Estriol high sensitive Saliva ELISA (Cat. No. 30121046) can be adapted on different ELISA processors. Open automated platforms like the EVOlyzer® from Tecan have been tested and show good correlation to the manual runs:
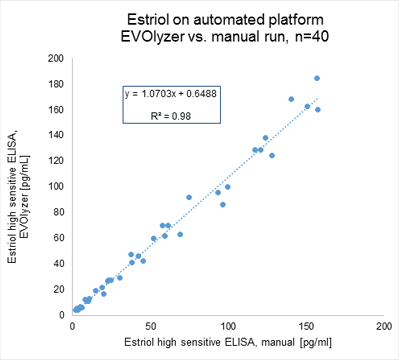
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. CE IVD under IVDR to be launched soon. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.