In the product range of tropical diseases IBL International now offers an assay for diagnosis of Zika virus infection, a dramatically spreading disease in the Americas and Pacific region
Zika virus, a single-stranded RNA virus of the Flaviviridae family, is an emerging mosquito-borne virus caused by the bite of an infected Aedes mosquito. In May 2015, the public health authorities of Brazil confirmed the transmission of Zika virus in the country’s northeast. Since October 2015, other countries and territories of the Americas have reported the presence of the virus. The World Health Organization has declared the Zika virus an international public health emergency, prompted by growing concern that it could cause birth defects. As many as four million people could be infected by the end of 2016.
Symptoms are similar to those of dengue or chikungunya. Most common are skin rash, mild fever, conjunctivitis, and muscle pain. Pregnant women in general are at special risk because the infection appears to be linked to the development of unusually small heads and brain damage in newborns – called microcephaly – as well as other neurological defects.
Due to these facts an early diagnosis is of utmost importance. Aside from diagnosis on clinical symptoms and epidemiological circumstances, virological and serological blood tests are important to confirm infection.
IBL International has now launched an ELISA for the detection of IgM antibodies based on highly sensitive and specific µ-capture technique as an aid in the serologic diagnosis of Zika virus infection.
The assay features
- Diagnosis of Zika virus infection
- µ-capture technology
- No significant cross reactivity to other Flaviviridae
- Qualitative result evaluation
- Sensitivity > 99%, specificity 98.5%
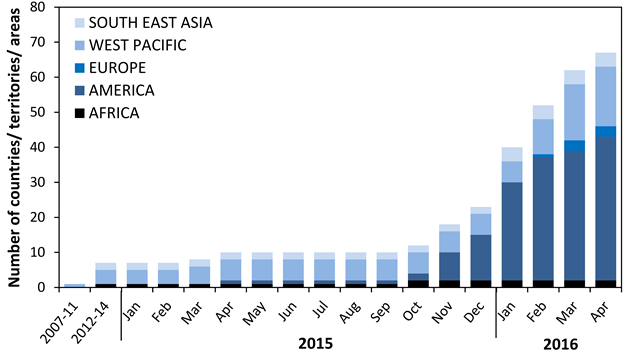
Cumulative number of countries, territories and areas by region reporting Zika virus transmission in years 2007-2016
Determination of the assay performance to discriminate between positive and negative clinical samples.
In total 140 samples:
9 positive Zika virus samples (BNI Hamburg, Germany)
47 negative samples (blood bank Frankfurt, Germany)
84 samples to check cross reactivity (United States, Uganda, Guadeloupe, New Caledonia)
| Defined sample status |
| positive | negative | ∑ |
| IBL Zika virus IGM µ-capture ELISA | positive | 9 | 0 | 9 |
| negative | 0 | 129 | 129 |
| ∑ | 9 | 129 | 138 |
The IBL Zika virus IgM µ-capture ELISA shows 98.6% (138/140) agreement between measured result and the pre-defined sample status.
Excerpt of the Instruction for Use
Principle of the assay
The qualitative immunoenzymatic determination of specific IgM-class antibodies is based on the ELISA (Enzyme-linked Immunosorbent Assay) μ-capture technique.Microplates are coated with anti-human IgM-class antibodies to bind the corresponding antibodies of the sample. After washing the wells to remove all unbound sample material a horseradish peroxidase (HRP) labelled Zika virus antigen is added. This antigen-conjugate binds to the captured specific IgM antibodies. In a second washing step unbound conjugate is removed. The immune complex formed by the bound conjugate is visualized by adding Tetramethylbenzidine (TMB) substrate which gives a blue reaction product. The intensity of this product is proportional to the amount of specific IgM antibodies in the sample. Sulphuric acid is added to stop the reaction. This produces a yellow endpoint colour. Absorbance at 450/620 nm is read using an ELISA microwell plate reader.
For concrete data please consult the Instruction for Use in the download box on the top right side.













