25-OH-Vitamin D ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 90min., 1 x 30min., 1 x 15min.
- Standard range
- 0 - 120 ng/mL
- Specimen / Volumes
- 25µL serum
- Substrate / isotope
- TMB 450nm
Enzyme immunoassay for the quantitative measurement of total 25-OH-Vitamin D (25-OH-Vitamin D2 and 25-OH-Vitamin D3) in human serum as a physiological marker as an aid to the diagnosis of vitamin D sufficiency. Patient population includes individuals who are at risk to deviate from sufficient levels of vitamin D. The 25-OH-Vitamin D ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. The assay is adaptable by laboratory personnel to automate on open ELISA based liquid handler platforms; however, the programming of the steps and timing required by the manual kit assay test instructions must be strictly adhered to and verified by the laboratory. Test results are calculated from a standard curve and compared to defined cut-off. The test kit is intended for professional laboratory use by trained personnel. The test kit is not for home or lay person use.
The accurate assessment of a person’s vitamin D status relies on the measurement of 25-OH-vitamin D total (25-OH-vitamin D2 + 25-OH-vitamin D3) levels. Therefore, our assay has been developed to detect both D2 and D3 variants. Balanced levels of vitamins in the human body are important to prevent disease (e.g. cardiovascular diseases, type 2 diabetes)[3]. Among essential vitamins, vitamin D is important to maintain calcium metabolism and bone health[2]. Additionally, vitamin D has received interest for its role in non-skeletal diseases and disorders[4,5]. It has been found that a large fraction of patients encountered in clinical practice are deficient in vitamin D[1]. Identifying patients with insufficient vitamin D levels in patient populations at risk from hypovitaminosis D, can help to maintain health and prevent disease[2].
| Suggested reference values[1,2] | |
| Deficiency | 0 - 20 ng/mL |
| Insufficiency | 21 - 29 ng/mL |
| Sufficiency | 30 - 79 ng/mL |
| Toxicity | >80 ng/mL |
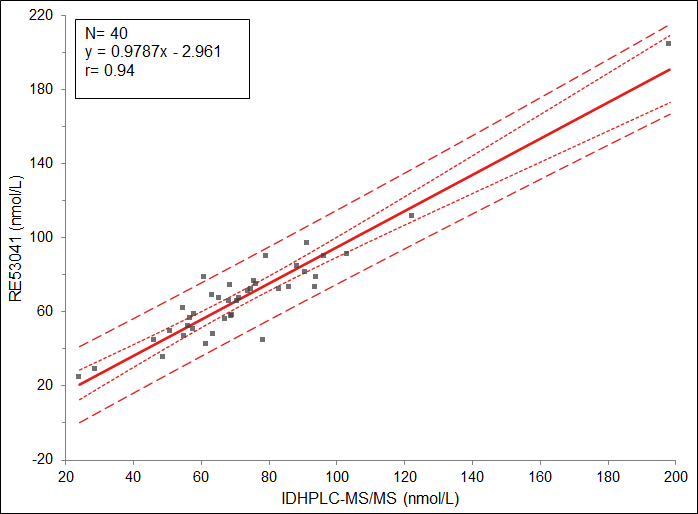
The Tecan-IBL 25-OH-Vitamin D ELISA is correlated against the IDHPLC-MS/MS reference method (The method is recognized by the Joint Committee for Traceability in Laboratory Medicine (JCTLM) as a reference measurement procedure (RMPs) of a higher-order).
The correlation was performed on 40 serum samples (CDC phase I samples, single-donor sera) comparing the results of TECAN-IBL 25-OH-Vitamin D ELISA to reference concentrations assigned by CDC using IDHPLC-MS/MS. The regression analysis showed a slope of 0.9787, an intercept of -2.961 nmol/L (nmol/L = 2.5 x ng/mL) with a correlation of r=0.94.
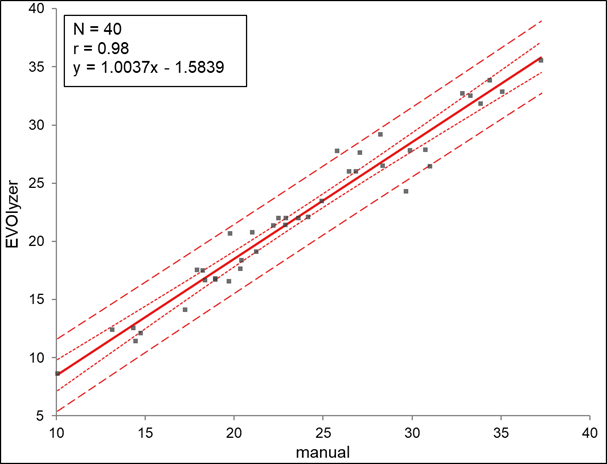
The Tecan-IBL 25-OH-Vitamin D ELISA has been developed to be adaptable to open ELISA processors. An in-house correlation has been performed comparing the manual work-up of the 25-OH-Vitamin D ELISA and the automated work-up of the 25-OH-Vitamin D ELISA on the TECAN Freedom EVOlyzer®.
Advantages of our ELISA
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.