17-Hydroxy-Progesterone (17-OHP) ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 60 min, 1 x 30 min
- Standard range
- 0.15 - 20 ng/mL
- Specimen / Volumes
- 25 µL serum, plasma
- Substrate / isotope
- TMB 450 nm
Intended Purpose
Enzyme immunoassay for the in-vitro-diagnostic quantitative determination of 17-OH-Progesterone in human serum and EDTA plasma from women, men, and children. This test is intended as an aid to diagnosis and monitoring of congenital adrenal hyperplasia, in which 17-OH-Progesterone levels are found to be elevated in comparison with a corresponding population of apparently healthy individuals due to genetic 21-hydroxylase deficiency. Additionally, other clinical observations such as physical exams, quantification of other steroidal hormones (androstenedione and cortisol) and genetic testing are employed to assess the status of congenital adrenal hyperplasia. This test is intended as an aid to diagnosis and monitoring of late-onset congenital adrenal hyperplasia, in which 17-OH-Progesterone levels are found to be elevated in comparison with a corresponding population of apparently healthy individuals due to perturbations (21-hydroxylase deficiency, or androgen “backdoor” pathway) in the metabolism of 17-OH-Progesterone that leads to the accumulation of this steroid. Additionally, other clinical observations such as physical exams, quantification of other steroidal hormones (androstenedione, cortisol, and dihydrotestosterone), and genetic testing are employed to assess the status of late-onset congenital adrenal hyperplasia. In certain cases of congenital adrenal hyperplasia, 17-OH-Progesterone levels are quantified before and after stimulation with ACTH. The 17-OH-Progesterone ELISA is NOT appropriate for use in neonatal screening and for monitoring patients with hypogonadotropic hypogonadism.
The 17-OH-Progesterone ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortex mixer and pipettes to execute the test.The assay is adaptable by laboratory personnel to automate on open ELISA based liquid handler platforms; however, the programming of the steps and timing required by the manual kit assay test instructions must be strictly adhered to and verified by the laboratory. Test results may be calculated manually from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges). The test kit is intended for professional laboratory use by trained personnel. The test kit is not for home or lay person use.
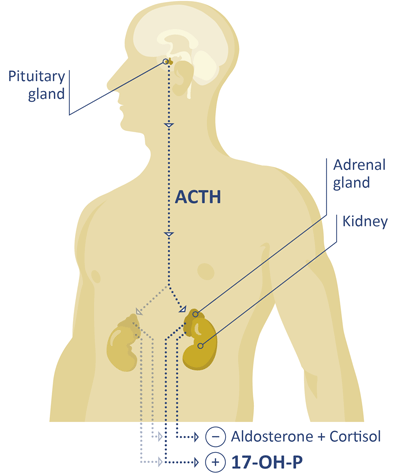
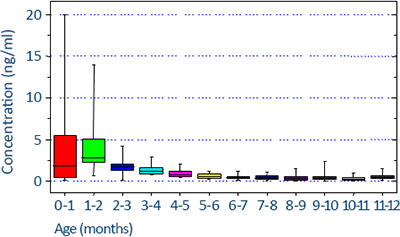
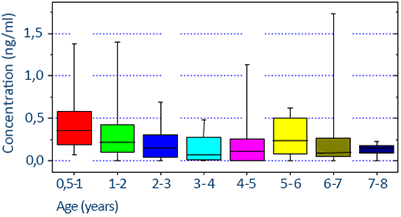
Clinical significance
17-Hydroxy progesterone (17-OHP) is a steroid hormone, which can be found in circulation in the human body. The role of 17-OHP as an intermediate in the biosynthesis of cortisol is well established in the peer-reviewed scientific literature.[2]
Quantification of 17-OHP from serum or plasma can be used as in aid in the diagnosis or monitoring of congenital adrenal hyperplasia (CAH) and non-classical congenital adrenal hyperplasia (NCCAH). These conditions are associated with deficiencies in the activity of 21-hydroxylase. This enzyme metabolizes 17-OHP in the pathway to cortisol. The identification of 17-OHP as a biomarker for CAH and NCCAH is consistent with the blockade of the biochemical pathway of cortisol production. In the perturbed biosynthesis, 17-OHP is the last intermediate before the dysfunctional enzyme (21-hydroxylase, P450c21). An increase of concentration is the consequence of the consistent influx by the conversion of progesterone and 17-OH pregnenolone to 17-OHP. Healthy patients can be identified when their 17-OHP levels are within the respective reference ranges.[1]
17-OHP levels are subject to variation. Samples should be taken in the early morning for all patients. A special requirement exists for menstruating women; their samples should be taken in the follicular phase. 17-OHP levels that were quantified under these restrictions are considered as “baseline” levels.
Patients that are affected by CAH or NCCAH can be identified with 17-OHP levels above the respective reference ranges. Clinicians should confirm high levels of 17-OHP that may indicate CAH, e.g. with the use of the gold-standard method (LC-MS), by examination of 17-OHP levels after cosyntropin stimulation or genetic testing.
Monitoring of patients with CAH is not solely dependent on 17-OHP levels but also includes the examination of other hormones. The aim of medical treatment is to suppress excess of androgenic hormones.[4] Consequently, periodically measuring androgen (testosterone, androstenedione) levels is recommended.[3]
For the monitoring of NCCAH patients the values acquired during analysis may not necessarily be viewed in correlation to a reference range but in perspective of the patient history of the affected individual.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.