Testosterone Saliva ELISA
- Regulatory Status
- EU: CE IVDR
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 2 h, 1 x 30 min
- Standard range
- 10 - 900 pg/mL
- Specimen / Volumes
- 50 µL saliva/without extraction
- Substrate / isotope
- TMB 450 nm
Enzyme immunoassay for the quantitative determination of free testosterone in human saliva.
The Testosterone Saliva ELISA is intended for the measurement of testosterone in saliva in adults. Measurement of testosterone has been shown to be useful in the evaluation of hormone imbalance that results in various clinical conditions like hypogonadism (androgen deficiency) in men and hypergonadism (excess of androgen hormones) e.g. polycystic ovaries in women.
The Testosterone Saliva ELISA is a solid phase enzyme-linked immunosorbent assay (ELISA), based on the principle of competitive binding and measured on an absorbance reader. The assay is semi-automated requiring general purpose laboratory instruments and consumables such as absorbance microplate reader/washer, vortexer and pipettes to execute the test. The assay is adaptable by laboratory personnel to automate on open ELISA based liquid handler platforms; however, the programming of the steps and timing required by the manual kit assay test instructions must be strictly adhered to and verified by the laboratory. Test results may be calculated from a standard curve and compared to laboratory established reference ranges from healthy adults (i.e. normal ranges).
The test kit is intended for professional laboratory use by trained personnel. The test kit is not for home or layperson use. The Testosterone Saliva ELISA is NOT intended for point-of-care settings.
The improved IBL International Testosterone Saliva ELISA is calibrated to the LC-MS/MS reference method and includes interchangeable reagents within the IBL saliva ELISA product line.
The Testosterone Saliva ELISA can easily be automated on open platforms like the EVOlyzer® from Tecan.
Benefits of the Testosterone Saliva ELISA
Mass spectrometry (MS) is considered as the reference method for steroid hormone quantification. This methodology does however represent a large investment and as such is not available to all laboratories. All the IBL saliva assays have been developed to correlate to this reference method, and even more importantly, only require low sample volume for quantification of the steroid hormones.

Testosterone is a C19-steroid and plays an important role in the human body. It is also considered a pleiotropic hormone [1], meaning it affects different phenotypes and plays an important role in the human body. In men testosterone is produced by the testes as well minimally by the adrenal cortex. While in women the testosterone level is secreted from the adrenal cortex (25%) and the ovarian stroma (25%). The remaining testosterone production occurs in peripheral tissues such as bone, breast, muscle and fat [2]. In healthy individuals the testosterone level follows a diurnal rhythm with a peak in the morning hours and low levels throughout the day [3]. In the circulation, the main part of testosterone is bound to plasma proteins like sex hormone binding globuline (SHBG) and albumin. Only 1 – 2% of the testosterone is unbound and therefore biologically active [4;5].
The free testosterone is released via the salivary glands, which is clinically relevant [8]. Nevertheless, the concentration of testosterone in saliva reflects the level of free testosterone in plasma [4, 6, 7].
As mentioned above testosterone is no exception within the group of steroid hormones as concentrations follow a diurnal rhythm [9], thus, sampling time and number of samples is crucial in a clinical setting to reliable evaluate the peak and baseline level of testosterone individually.
Measurement in men is used to diagnose clinical hypogonadism, while in women it is applied to help diagnose hyperandrogenic states e.g., polycystic ovarian syndrome (PCOS), hirsutism (excessive hair) [10], ovarian failure or virilization (masculinization) [11].
Our Saliva ELISA product line (Cortisol, Testosterone, Progesterone, DHEA) can be processed in a similar way and be easily performed in combination:
*Valid lot numbers of TMB, stop solution and wash solutionare stated on the kits outer labels and Quality Controlcertificates.
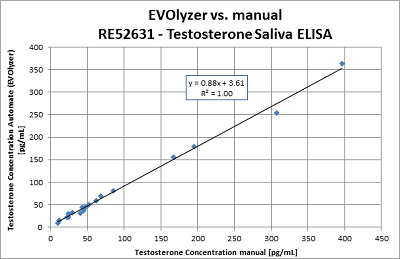
The IBL International Testosterone Saliva ELISA (RE52631) can be adapted on different ELISA processors. Open automated platforms like the EVOlyzer® from Tecan have been tested and show good correlation to the manual runs:
Testosterone is a steroid hormone, produced in the testes of males and in smaller quantities in the ovaries of women. In both men and women a substantial amount is produced by the conversion of circulating DHEA-S, DHEA and androstenedione. It shows a diurnal rhythm, with highest levels in the morning and a nadir around midnight.
Testosterone plays an important role in the development of male reproductive tissues, supports increased muscle, bone mass, and hair growth and is essential for health, stamina, sexual function, cardiovascular health, and a healthy immune system.
The measurement of testosterone is used in the diagnosis and the treatment of disorders involving androgens, the male sex hormones.
These include primary and secondary hypogonadism, disorders originating from testicular problems or defects of the hypothalamus or pituitary glands. Furthermore with impotence in males, in female hirsutism (excessive hair), virilization (masculinization) due to tumors, polycystic ovaries and adrenogenital syndroms. Testosterone together with Cortisol levels also represents a useful parameter in stress research and sports medicine.
Only 1-5% of the hormone Testosterone is unbound from plasma proteins and therefore biologically active. It is released via the salivary glands and mainly transformed to 5a-dihydrotestosterone. The concentration of testosterone in saliva, reflects however the level of free testosterone in plasma.
Testosterone is no exception within steroid hormones as concentrations follow a diurnal rhythm. Thus, sampling time and number of samples is crucial in a clinical setting to reliable evaluate the peak and baseline level of testosterone individually.
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. CE IVD under IVDR to be launched soon. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
For concrete data please consult the Instruction for Use in the download box on the top right side.
1. Tygi, V., Scordo, M., Yoon, R. S., Liporace, F. A., & Greene, L. W. (2017). Revisiting the role of testosterone: Are we missing something?. Reviews in urology, 19(1), 16.
2. Burger, H. G. (2002). Androgen production in women. Fertility and sterility, 77, 3-5.
3. Goncharov, N., Katsya, G., Dobracheva, A., Nizhnik, A., Kolesnikova, G., Herbst, V., & Westermann, J. (2006). Diagnostic significance of free salivary testosterone measurement using a direct luminescence immunoassay in healthy men and in patients with disorders of androgenic status. The Aging Male, 9(2), 111-122.
4. David Wild. The Immunoassay Handbook. Theory and Applications of Ligand Binding, ELISA and Related Techniques. 4th Edition. 2013, pp 722, 731-733, 746, 748-749.
5. Shea, J. L., Wong, P. Y., & Chen, Y. (2014). Free testosterone: clinical utility and important analytical aspects of measurement. Advances in clinical chemistry, 63, 59-84.
6. Lewis, J.G. Steroid Analysis in Saliva: An overview. Clin Biochem Rev 2006, Volume 27:139-146
7. Goncharov N, Katsya G, Dobracheva A, Nizhnik A, Kolesnikova G, Herbst V, Westermann J. 2006. Diagnostic significance of free salivary testosterone measurement using a direct luminescence immunoassay in healthy men and in patients with disorders of androgenic status. Aging Male. 9(2): 111-22.
8. Brambilla, D. J., Matsumoto, A. M., Araujo, A. B., & McKinlay, J. B. (2009). The effect of diurnal variation on clinical measurement of serum testosterone and other sex hormone levels in men. The Journal of Clinical Endocrinology & Metabolism, 94(3), 907-913.
9. Wood, P. (2009). Salivary steroid assays–research or routine?. Annals of clinical biochemistry, 46(3), 183-196.
10. Škrgatić, L., & Trgovčić, I. (2013). Hyperandrogenemia association with acne and hirsutism severity in Croatian women with polycystic ovary syndrome. Acta dermatovenerologica Croatica: ADC, 21(2), 105-112.
11. Lothar Thomas. Labor und Diagnose. Indikation und Bewertung von Laborbefunden für die medizinische Diagnostik. Band 2. 2012 Chapter 34, pp. 1841-1842.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.