Cortisol Saliva ELISA
- Regulatory Status
- IVD
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 1 x 2 h, 1 x 30 min
- Standard range
- 0.15 - 30 ng/mL
- Specimen / Volumes
- 50 µL saliva
- Substrate / isotope
- TMB 450nm
The IBL International Cortisol Saliva ELISA kit is an excellent alternative to the current complex and relatively expensive LC-MS/MS method to determine cortisol in saliva as accurately as possible.
Advantages of the IBL Cortisol Saliva ELISA
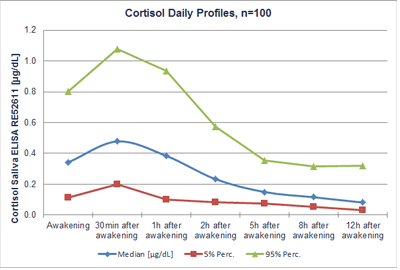
Diurnal rhythm
The normal cortisol levels in human saliva depend on the time of day. The timing of the cortisol peak, however, does not depend on the actual time of day or daylight, but correlates with the usual wake-up time. The new reference ranges have been carefully determined on the basis of 100 day profiles. Starting with batch no. ECO137, the following reference values have to be considered:
| Time after awakening(h) | Cortisol (saliva) Range - RE52611 | |||||
| (♂/♀; > 6y; n= 100; 5%-95% percentile) | ||||||
| Median | Range (µg/dL) | Median | Range (nmol/L) | |||
| (µg/dL) | 5% | 95% | (nmol/L) | 5% | 95% | |
| Awakening | 0.343 | 0.113 | 0.803 | 9.47 | 3.12 | 22.17 |
| 0.5 | 0.478 | 0.200 | 1.076 | 13.19 | 5.52 | 29.70 |
| 1 | 0.384 | 0.101 | 0.936 | 10.60 | 2.79 | 25.82 |
| 2 | 0.234 | 0.083 | 0.574 | 6.44 | 2.29 | 15.85 |
| 5 | 0.150 | 0.074 | 0.355 | 4.14 | 2.04 | 9.79 |
| 8 | 0.116 | 0.055 | 0.314 | 3.20 | 1.53 | 8.67 |
| 12 | 0.082 | 0.032 | 0.322 | 2.26 | 0.87 | 8.89 |
Sensitive and reliable
The investigation of cortisol in saliva is indicated in the diagnostic work-up of clinical issues in various scientific/medical fields, particularly endocrinology, psychology, sports medicine, pediatrics, anti-aging medicine and veterinary medicine
Measuring the free cortisol levels in saliva is a very simple and convenient way to check the function of the adrenal gland. Persistently elevated or decreased cortisol levels are often found in patients with adrenal dysfunction. Adrenal dysfunction can occur in many other diseases, such as fibromyalgia, hypothyroidism, chronic fatigue syndrome and premature menopause.
The salivary cortisol test can also be used in patients with conditions such as reduced stress tolerance, fatigue, sleep disorders and depression.
Standardized reagents
Our Saliva ELISA product line has been revised so that the main steroids (cortisol, testosterone, progesterone, DHEA) may be processed in a similar way:
Thanks to the similar processing and assembling, the assays can easily be performed in combination.
*Product availability and regulatory status may vary across regions outside the EU depending on local country-specific registration. Consult with your Tecan associate for further information.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.