IGFBP-3 ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 2 x 1 hr,1 x 30 min
- Standard range
- 0.4 -30 ng/mL
- Specimen / Volumes
- 10 µl Serum, Plasma
- Substrate / isotope
- TMB, 450 nm
This enzyme immunoassay kit is suited for measuring IGFBP-3 in human serum, EDTA and Heparinplasma for diagnostic and scientific purposes.
Insulin-like growth factors (IGF)-I and -II are bound to specific binding proteins (IGFBPs) in the circulation. To date, at least six binding proteins can be distinguished on the basis of their amino acid sequence. They are designated as IGFBP-1, IGFBP-2, ... IGPBP-6 (1). The predominating IGFBP in blood is IGFBP-3, which largely determines the total IGF-I and IGF-II concentration. In contrast to the other binding proteins, IGFBP-3 has the property to associate with an acid-labile subunit (ALS) after binding of either IGF-I or IGF-II (3-5). Most of the IGFBP-3 in plasma is present as high molecular weight ternary complex, however, small amounts of free IGFBP-3 are also found (6,7).
The development of a specific immunoassays for IGFBP-3, which detects IGFBP-3 in the ternary complex, provided new in-sights into IGFP-3 regulation (6-9). On the basis of these findings serum IGFBP-3 has been proven to be an additional useful test in the repertoire of diagnostic tools for evaluation of growth disorders (7,8).
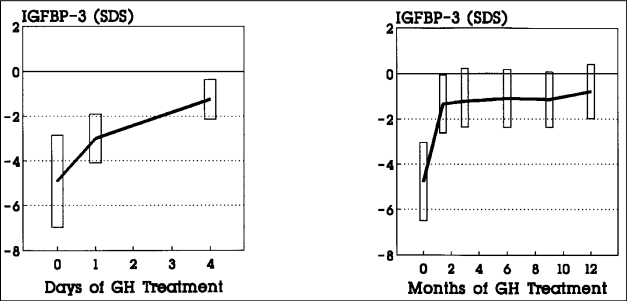
Several factors besides GH influence IGFBP-3 levels: age including sexual development, nutrition, hypothyroidism, diabetes mellitus, liver function and kidney function. IGFBP-3 levels are decreased by malnutrition, although less than IGF-I, in hypothyroidism, in diabetes mellitus and in hepatic failure (6-8), but are increased in chronic renal failure (6,10,11). Measurement over 24 hours revealed no circadian rhythm (12,13). For clinical practice, the most important regulatory factor is GH. Single IGFBP-3 measurements correlate significantly with the logarithm of the integrated spontaneous GH secretion (8,14). In patients with GH deficiency, IGFBP-3 levels are subnormal and increase gradually to within the normal range after several days of GH administration (7,8). The slow response to GH and constant circadian levels during chronic daily application of GH (13) suggest that IGFBP-3 reflects the GH secretory state over days.
The major advantages of IGFBP-3 over IGF-I are:
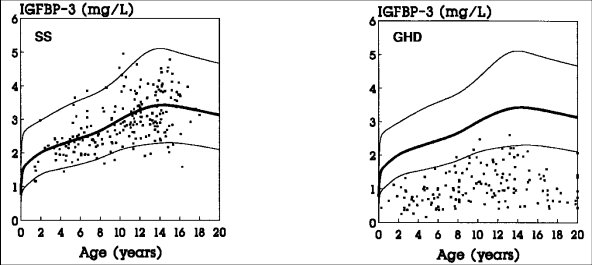
Fig. 1: Serum IGFBP-3 levels in patients with short stature without GH deficiency (SS: constitutional delay of growth and adolescence, familial short stature, intra-uterine growth retardation) and in idiopathic or organic GH deficiency (GHD). The normal range is given by the 5th, 50th and 95th percentile.
Fig. 2: IGFBP-3 levels in GH deficient children before and during GH treatment. Because of the agedependence, values are given as the mean of standard deviation scores (SDS).
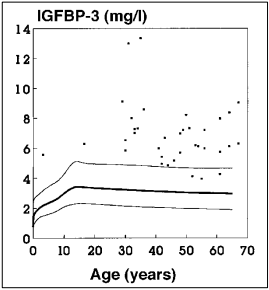
In normal tall children and adolescents without excessive GH secretion or in patients with Sotos syndrome, IGFBP-3 levels are normal or slightly increased. In contrast, children with pituitary gigantism or adults with acromegaly have clearly elevated levels (Figure 3) (6,15) that normalize on successful treatment. Therefore, IGFBP-3 is also a useful parameter for the detection of excessive GH secretion and monitoring therapy efficacy. In precocious puberty, IGFBP-3 levels are clearly increased by chronological age, whereas patients with premature thelarche have IGFBP-3 levels in the upper normal range (15).
Fig. 3: Serum IGFBP-3 levels in acromegaly. The normal range is given by the 5th, 50th and 95th percentile.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.