GLP-1 (active forms) ELISA
- Regulatory Status
- RUO
- Kit size
- 12 x 8
- Method
- ELISA
- Incubation time
- 2 x 1h, 1 x 30 min
- Standard range
- 1.25 - 80 pmol/L
- Specimen / Volumes
- 100µL EDTA plasma
- Substrate / isotope
- TMB 450 nm
| instructions for use |
|---|
| instructions for use |
|---|
The gut hormone glucagon-like peptide-1 (GLP-1) is secreted in response to nutrient intake and plays an essential role in the regulation of glucose homeostasis. There are multiple forms of GLP-1, among which GLP-1(7-37) and GLP-1(7-36)NH2 are considered to be biologically active. Accurate measurement of both forms of active GLP-1 is not only important for understanding the physiological mechanisms of blood glucose regulation in healthy people and type 2 diabetes patients, but it is also of utmost importance for both clinical and preclinical studies of GLP-1 agonists and DPP4 inhibitors (e.g. Sitagliptin®, Saxagliptin®).
Among the currently available methods for measuring active GLP-1 peptides, immunoassays are greatly favored because of their sensitivity. However, most immunoassays still require an extraction step of active GLP-1 which is tedious and time-consuming.
To meet these needs, a reliable and user-friendly assay with a high sensitivity is essential due to low plasma levels of the active form GLP-1.
Advantages of our assay
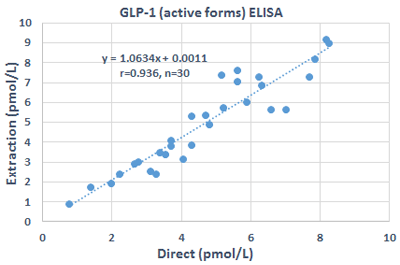
IBL International is happy to present the GLP-1 active forms ELISA without extraction for the reliable measurement of biologically active forms of GLP-1.
Our assay needs no extraction step and detects both forms of the active GLP-1 with high sensitivity.
* distributed by IBL International
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.