hTau total ELISA
- Regulatory Status
- EU: CE
- Kit size
- 12x8
- Method
- ELISA
- Incubation time
- 1x2h, 1x90min, 1x30min
- Standard range
- 25 - 1000 pg/mL
- Specimen / Volumes
- 25 µL CSF
- Substrate / isotope
- TMB 450 nm
Along with amyloid-β plaques, the formation of tau fibrils within the nerve cells, particularly in the cerebral cortex and notably in the limbic lobe, is another pathological hallmark of Alzheimer's disease. The concentration of Tau in the CSF is a marker of neuronal cell death. In Alzheimer's disease, however, it increases only at a later stage of the disease when the patient already displays significant cognitive impairment. Therefore, the determination of total tau in CSF is important for the diagnosis of Alzheimer's disease in addition to the determination of amyloid β-peptides. However, increased tau levels are found in other neurodegenerative diseases as well. These disorders are generally referred to as tauopathies and include, inter alia, frontotemporal lobar degeneration (FTLD), Pick's disease and corticobasal degeneration (CBD). Tau levels are markedly elevated in Creutzfeldt-Jakob disease.
The hTAU total ELISA from Analytik Jena, manfuactured by AJ Roboscreen and distributed by IBL International, was optimized so that it can be processed in parallel with the IBL Amyloid-β CSF ELISA.
As the graph below shows, the hTAU total ELISA from Analytik Jena, manufactured by AJ Roboscreen and distributed by IBL International, correlates superbly with the INNOTEST ® hTAU.
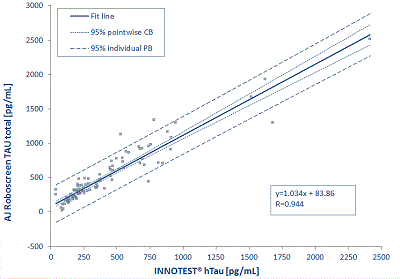
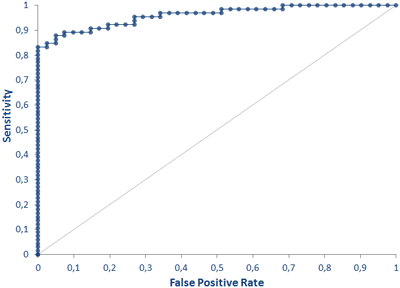
Clinical validation of the TAU total ELISA was performed at the University Hospital Erlangen in the CSF Lab of the Department of Psychiatry and Psychotherapy (Prof. Dr. Lewczuk). Tau levels in the cerebrospinal fluid of patients with Alzheimer's disease and mild cognitive impairment (collectively AD (n = 72) and control subjects (n = 41)) show highly significant differences between the groups.
Naturally, the TAU total ELISA, just like our Amyloid-β CSF ELISAs, displays an excellent analytical precision with an inter-assay variance of <5% (Table 1), intra-assay variance of <5% (Table 2) and inter- LOT and operator variance of <6% (Table 3).
| TAU total ELISA | ||
| Sample | MW | CV |
| [pg/mL] | [%] | |
| 1 | 182 | 4.7 |
| 2 | 315 | 4.4 |
| 3 | 343 | 4.3 |
| TAU total ELISA | ||
| Sample | MW | CV |
| [pg/mL] | [%] | |
| 1 | 179 | 4.5 |
| 2 | 307 | 2.7 |
| 3 | 338 | 2.1 |
| TAU total ELISA | ||
| Sample | MW | CV |
| [pg/mL] | [%] | |
| 1 | 180 | 5.7 |
| 2 | 311 | 4.4 |
| 3 | 342 | 5.6 |
Additional advantages of our ELISA are:
General Information
In 2010, the number of dementia patients worldwide was estimated at 36 million. Assuming an ongoing lack of sufficient preventive and curative treatments, this is expected to double every 20 years. Alzheimer’s Disease accounts for roughly 60‐70% of all dementia cases. Both prevalence and incidence increase with age. Prevalence is around 1% in those aged 65‐69, and more than 30% in those aged 90 or older. The first case of Alzheimer’s Disease was defined and reported in 1907 by German scientist Dr. Alois Alzheimer. He described two hallmarks of the Disease ‐ the plaques and tangles in the brains of Alzheimer’s patients. The plaques are formed by Amyloid-beta peptide deposits and the tangles are composed of hyperphosphorylated Tau proteins. Amyloidosis occurs as early as the preclinical stage. The first cognitive deficits can manifest themselves in the mild cognitive impairment (MCI) stage, while in the dementia stage patients are unable to do any work or daily chores. The concentration of TAU in CSF is therefore recognized as a one biomarker in diagnosing Alzheimer’s Disease. Moreover, a number of independent studies showed that combining TAU‐protein and amyloid‐beta (1‐42) levels will enhance specificity and sensitivity of diagnosis of Alzheimer’s Disease, which is further enhanced when combining TAU levels to Amyloid-beta ratio measurement.
For concrete data please consult the Instruction for Use in the download box on the top right side.
Our comprehensive immunoassay portfolio includes a number of specialty diagnostic immunoassays for endocrinology, immunology and autoimmunity, as well as for diagnosis of multiple infectious diseases. We are pioneers and market leaders in saliva diagnostics, with over 40 years of experience supplying a broad portfolio of luminescence- and ELISA-based tests, including our highly acclaimed HMGB1 and MuSK-Ab ELISAs.
And as experts in laboratory automation, we can support our customers with the protocols for open ELISA platforms, such as the Freedom EVOlyzer or Thunderbolt®.
All products are only available for sale to laboratory professionals and may not be available in all countries. Availability and regulatory status may vary across regions depending on local country-specific registration. Please always read and follow the instructions for use.
All of our assays have been designed and manufactured to meet the highest global regulatory requirements and quality standards. Tecan is certified under ISO 9001:2015, ISO 13485:2016 and is audited by a notified body according to Medical Device Single Audit Program (MDSAP).

As part of the Tecan Group, we have a leading market position in diagnostics and research, with over 40 years of experience in the development, manufacture and supply of enzyme-, radiolabel- and luminescence-based immunoassays.
Our range of high-quality immunoassays is supported by a diverse portfolio of automated solutions, making us the perfect partner for you and your customers.
At Tecan, we are driven to improve people’s lives and health.